Japan Patent Office publishes the Examination Handbook for Patent and Utility Model.
The section about Biological inventions is as follows.
Chapter 2 Biological Inventions
In this chapter, the application of Examination Guidelines to patent applications relating to biological inventions is explained.
Biological inventions are inventions relating to matters consisting of or comprising biological material, or processes of producing, treating or using the biological material.
See Examination Guidelines in relation to matters in description requirements and requirements for patentability which are not explained in this chapter.
Definition of Terms used in this Chapter
(i) Biological Materials;
Biological materials are materials which have genetic information, and can replicate or breed by themselves or can replicate in vivo on the basis of the genetic information. Namely, the biological materials include nucleic acids (genes, vectors, etc.), polypeptides (proteins, monoclonal antibodies, etc.), microorganisms (see (ii)), and animals and plants (see (iii) and (iv)).
(ii) Microorganisms;
Microorganisms include animal or plant cells (including stem cells, dedifferentiated cells and differentiated cells) and tissue cultures, in addition to fungi, bacteria, unicellular algae, viruses and protozoans. The microorganisms also include fused cells (including hybridomas) obtained by genetic engineering (see (v)), dedifferentiated cells and transformants (microorganisms).
(iii) Animals;
Animals mean animals obtained by classifying organisms other than microorganisms (see (ii)) into animals and plants. Animals include an animal itself, a part of the animal, and a fertilized egg. Animals also include a transformant (animal) obtained by genetic engineering (see (v)).
(iv) Plants;
Plants mean plants obtained by classifying organisms other than microorganisms (see (ii)) into animals and plants. Plants include a plant itself, a part of the plant, and a seed. Plants also include a transformant (plant) obtained by genetic engineering (see (v)).
(v) Genetic Engineering;
Genetic engineering means a technique of artificially manipulating genes by gene recombination, cell fusion or the like.
1. Requirements for Description
1.1 Enablement Requirement (Article 36(4)(i))
Determination on enablement requirement relating to biological inventions is carried out according to “Part II, Chapter 1, Section 1 Enablement Requirement” in Examination Guidelines.
1.1.1 Invention of a Product
An invention of a product must be clearly stated in the description. Further, the invention of a product must be stated in such a manner so as to enable a person skilled in the art to produce and use the product. However, this is not the case when a person skilled in the art can produce the product and use the product on the basis of the statements in the description and drawings as well as the common general knowledge at the time of filing.
If the means for producing a biological material cannot be stated in such a way that a person skilled in the art can produce the biological material, it is necessary to deposit the biological material in accordance with Article 27bis of Regulations under the Patent Act (for the details, see "1.1.4 Deposit and Furnishing of Biological Material").
In order to satisfy the enablement requirement in an invention relating to a biological material, for example, it may be described in the description as follows.
(1) Invention relating to Nucleic acids and Polypeptides
a Nucleic acids such as Genes
In order to show that a gene can be produced in an invention relating to the gene, a production process may be described, such as the origin or source, a condition of treatment, a process of collecting or purifying, and a means for identifying.
If genes are claimed in a generic form in the claim (if genes are specified by "deleted, substituted or added", "hybridized" or "having more than X% of sequence identity", etc.), in a case where it is necessary to make trials and errors, and/or complicated and sophisticated experimentation beyond the extent to which a person skilled in the art should be reasonably expected to obtain those genes, then such a description is not described in such a manner that enables a person skilled in the art to produce the product.
For example, if many genes which do not have the same function as the gene actually obtained are included in the genes whose sequence identity is extremely low, trials and errors, and/or complicated and sophisticated experimentation beyond the extent to which a person skilled in the art should be reasonably expected is generally needed to select the genes with the same function as said gene obtained from those genes. Therefore, in such a case, regarding the claimed invention which includes the gene actually obtained and gene whose sequence identity is extremely low to said gene obtained, and is specified by their function, the description is not described in such a manner that enables a person skilled in the art to produce the product.
Example 1: A polynucleotide selected from the group consisting of:
(i) a polynucleotide whose sequence is represented by ATGTATCGG......TGCCT
(ii) a polynucleotide whose DNA sequence has more than X% of sequence identity to that of (i) and which encodes the protein having the activity of enzyme B.
(Note) A protein encoded by the polynucleotide of (i) has the activity of enzyme B. X% represents extremely low identity.
(Explanation)
The polynucleotide of (ii) includes a polynucelotide whose sequence identity is extremely low in relation to the polynucleotide of (i) actually obtained which is described in the description, although the polynucleotide of (ii) is specified by its function. If "polynucleotide whose DNA sequence having more than X% of sequence identity to that of (i) and which encodes the protein having the activity of enzyme B" includes many polynucleotides which encode the protein not having the activity of enzyme B, trials and errors, and/or complicated and sophisticated experimentation beyond the extent to which a person skilled in the art should be reasonably expected is generally needed to select the polynucleotides which encode the protein having the activity of enzyme B from said polynucleotides. Therefore, the description is not described in such a manner that enables a person skilled in the art to produce the product.
In order to show how an invention relating to a gene can be used, it may be described that the gene has a specific function (the "specific function" here means a "function from which a specific use with technical meanings can be assumed"). For example, in a case of an invention relating to a structural gene, it may be described that a protein encoded by said gene has the specific function.
For example, if genes are claimed in a generic form and the function is not specified in the claim (genes are specified only by "deleted, substituted or added", "hybridized" or "having more than X% of sequence identity", etc.), the genes claimed in a generic form contain the ones which do not have said function and the part of said genes cannot be used. Therefore, in this case, the description is not described in such a manner that enables a person skilled in the art to use the product.
Example 2: A polynucleotide selected from the group consisting of:
(i) a polynucleotide whose DNA sequence is represented by ATGTATCGG......TGCCT
(ii) a polynucleotide whose DNA sequence identity has more than X% of identity to that of (i)
(Note) A protein encoded by the polynucleotide of (i) has the activity of enzyme B.
(Explanation)
Since the polynucleotide of (ii) is not specified by its function, the polynucleotide includes a polynucleotide encoding a protein not having the activity of enzyme B. Since the polynucleotide does not have the specific function, the description is not described in such a manner that enables a person skilled in the art to use the product.
b Proteins
In order to show that a recombinant protein can be produced in an invention relating to the recombinant protein, a production process, such as an acquiring means of genes encoding the recombinant protein, a process of transferring genes into microorganisms, animals or plants, a process of collecting the recombinant protein from transformants in which genes are introduced, and a means for identifying the recombinant protein, may be described. (see "1.1.1(1)a. Nucleic acids such as Genes" mentioned above for dealing with the enablement requirement if recombinant proteins are claimed in a generic form.)
In order to show that a protein can be produced in an invention relating to the protein obtained by isolating from or purifying natural products, a production process, such as an acquiring means of microorganisms producing the protein, a process of culturing the microorganisms, and a process of isolating or purifying the protein, may be described.
c Antibodies
In order to show that an antibody can be produced in an invention relating to the antibody, a production process, such as an acquiring or producing means of antigens, and a process of immunizing may be described. Especially, in a case of an invention relating to a monoclonal antibody, a production process, such as an acquiring or producing means of antigens, a process of immunizing, a process of selecting and collecting antibody-producing cells, and a means for identifying the monoclonal antibody, may be described.
(2) Invention relating to Microorganisms, Animals and Plants
a Fused Cells
In order to show that a fused cell can be produced in an invention relating to the fused cell, a production process, such as pre-treatment of parent cells, a condition of fusion, a process of selecting and collecting the fused cell, and a means for identifying the fused cell, may be described.
b Dedifferentiated Cells
In order to show that a cell produced by dedifferentiating a differentiated cell such as an induced pluripotent stem cell (iPS cell) can be produced in an invention relating to the dedifferentiated cell, a production process, such as a factor contributing to dedifferentiation of the differentiated cell (reprogramming factor), a species of cell in which the reprogramming factor is introduced, a process of introducing the reprogramming factor, a condition of culturing the cell in which the reprogramming factor is introduced, a process of selecting the dedifferentiated cell, and a means for identifying the dedifferentiated cell, may be described.
c Transformants
In order to show that a transformant can be produced in an invention relating to the transformant, a production process, such as a gene (or vector) to be introduced, an organism to which the gene (or vector) is introduced, a process of introducing the gene (or vector), a process of selecting and collecting the transformant, and a means for identifying the transformant, may be described.
d Microorganisms (obtained by means other than genetic engineering)
In order to clearly explain an invention relating to a fungus or bacterium, for example, a generic (species) name with nomenclature of fungi or bacteria, or a strain name in which the generic (species) name is added, may be described. In relation to an invention of a new strain, the characteristics of the strain as well as the difference (microbiological characteristics) between the new strain and the publicly known strains within the same species to which the new strain belongs may be described. In relation to an invention of a new genus (species), the taxonomic characteristics such as fungi and bacteria may be described in detail, and the reason why the microorganism is decided to be a new genus (species) may be described. Namely, the difference between the genus (species) and the existing similar genus (species) may be clearly described, and grounds on the decision may be described. The taxonomic characteristics may be described with reference to “Bergey’s Manual of Determinative Bacteriology” etc.
In order to show that a fungus or bacterium can be produced in an invention relating to the fungus or bacterium, a production process, such as a screening means and a mutagenesis means, may be described.
In order to clearly explain an invention relating to an animal or plant cell and show that the cell can be produced, the name of organism which is an origin of the cell may be described using the scientific name or standard Japanese name in accordance with zoological or botanical nomenclature, in principle. A combination of a specific gene or membrane protein of the animal or plant cell and the characteristics of the animal or plant cell, may be described. In order to show that the cell can be produced, a production process, such as a screening means and a mutagenesis means, may be described.
e Animals and Plants (obtained by means other than genetic engineering)
In order to show that an animal or plant per se and a part of the animal or plant can be produced in an invention relating to the animal or plant and a part of the animal or plant, production processes including species of a parent animal or plant, and a process of selecting the animal or plant of interest on the basis of objective indicators may be described step by step as production method.
As an objective indicator of an animal which is used to clearly explain an invention relating to the animal, numeric values or the like actually obtained by measuring the animal may be specifically described, and the characteristics may be described in comparison with those of publicly known animals, if necessary.
On the other hand, as an objective indicator of a plant which is used to clearly explain an invention relating to the plant, for example, the invention can be described not by a mere statement that the plant is high-yielding, but specific numeric values commonly used in conventional yield surveys, such as total number of fruits produced per stock, total weight of fruits produced per stock, and gross yield per are, etc., may be described, and the characteristics may be described in comparison with those of publicly known plants, if necessary.
Colors, such as leaf color, fruit color, and flower color may be expressed in accordance with official standards, such as the color atlas JIS Z8721 which is a specification of colors according to their three attributes, JIS Z8102 concerning color names and the R.H.S. color chart.
If the typical properties of the produced animal or plant cannot be expressed by a conventional breeding and cultivation processes which a person skilled in the art usually conducts, or if the properties are expressed only in specific environments or under specific breeding and cultivation processes, such specific breeding and cultivation processes should be specifically described.
1.1.2 Invention of a Process
An invention of a process must be clearly explained in the description of the invention. Further, description must be stated such that a person skilled in the art can use the process. However, this is not the case when a person skilled in the art can use the process on the basis of the statements in the description and drawings as well as the common general knowledge at the time of filing.
Relating to a "product" used in the invention of a process, see "1.1.1 Invention of Product."
If deposit of biological material is necessary, see "1.1.4 Deposit and Furnishing of Biological Material".
1.1.3 Invention of a Process for Producing a Product
An invention of a process of producing a product must be clearly explained in the description. Further, the invention of a process must be stated in such a manner so as to enable a person skilled in the art to produce the product by using the process.
However, this is not the case when a person skilled in the art can produce the product by the process based on the statements in the description and drawings as well as the common general knowledge at the time of filing.
If deposit of biological material is necessary, see "1.1.4 Deposit and Furnishing of Biological Material."
1.1.4 Deposit and Furnishing of Biological Material
In relation to an invention of biological material, if the description is not stated in such a manner that enables a person skilled in the art to produce the biological material, it is necessary to deposit the biological material in accordance with Article 27bis of Regulations under the Patent Act relating to deposition of microorganisms (see 1.1.4 (1)a). The deposited biological material is furnished in accordance with Article 27ter of Regulations under the Patent Act relating to furnishing of microbiological samples (see 1.1.4 (1) b).
If deposit of biological materials other than microorganisms such as genes, vectors, recombinant proteins, monoclonal antibodies, animals and plants is necessary, see "1.1.4 (2) Deposit of Genes, Vectors, Recombinant Proteins, Monoclonal Antibodies, Animals and Plants, etc.".
(1) Deposit and Furnishing of Microorganisms a Article 27bis of Regulations under the Patent Act (Deposition of Microorganisms)
A person desiring to file a patent application for an invention relating to a microorganism (hereinafter referred to as "an applicant"), shall deposit the microorganism with a depositary institution designated by the JPO Commissioner (hereinafter referred to as "designation of the institution"), an institution that has received a designation or another certification which is equivalent to a "designation of the institution" by a country that is not a party to the Budapest Treaty (the country is limited one which allows Japanese nationals to perform a procedure of the deposit of a microorganism for the purposes of patent procedure under the same conditions as in Japan and that is designated by the JPO Commissioner) or International Depositary Authorities (hereinafter, these are referred to as "depositary institution for the purposes of patent procedure"), unless a person skilled in the art can easily obtain the microorganism, shall state the accession number in the originally attached description, and shall attach a document certifying the fact that the microorganism has been deposited (hereinafter referred to as a "copy of the Receipt of an Original Deposit") to the request for the application.
The depositary institution designated by the JPO Commissioner issues a Receipt of a Reception immediately after receiving the application of patent depositary, and then issues a Receipt of an Original Deposit after testing the viability and finding the microorganism to be viable. As a Receipt of a Reception is not a document certifying the fact that the microorganism has been deposited, provided in Article 27bis of Regulations under the Patent Act, a Receipt of a Reception should not be attached to the request.
As the viability test of the microorganism takes time, an applicant may file a patent application stating a reference number which is written in a Receipt of a Reception, in the originally attached description. In this case, the applicant shall submit a copy of the Receipt of an Original Deposit immediately after the Receipt of an Original Deposit is issued.
When the Receipt of an Original Deposit is issued, the date of the original deposit shall be the date on which the microorganism was received by the depositary institution designated by the JPO Commissioner. Therefore, the application is not treated as having been deposited from the received date, if the depositary institution designated by the JPO Commissioner could not find the microorganism to be viable in the viability test, and did not issue the Receipt of an Original Deposit.
When a new accession number is assigned to the microorganism after filing the patent application, for the reason that, e.g., re-deposit was made, samples of the microorganism were transferred to another International Depositary Authority or the deposit was converted from the deposit under the national act to that under the Budapest
Treaty, the applicant or the patentee shall give a notice to that effect to the JPO Commissioner without delay. If a microorganism which was deposited with a depositary institution designated by the JPO Commissioner and was confirmed to be viable by the depositary institution is found to be no longer viable, the depositor, upon receipt of the "Notice that the microorganism cannot be furnished" from the depositary institution, should immediately deposit the same microorganism as that originally deposited. The applicant or the patentee should give a notice to that effect to the JPO Commissioner. In such a case, the newly deposited microorganism is treated as having been deposited without intermission since the original deposit was made.
b Article 27ter of Regulations under the Patent Act (Furnishing of Microbiological Samples)
The deposited microorganism can be furnished simultaneously with the registration for establishment of a patent right. Even prior to the registration for establishment of a patent right, in a case coming under Article 27ter(1)(ii) or (iii) of Regulations under the Patent Act, the microorganism can be furnished.
The deposit of a microorganism should be maintained at least during the duration of a patent right for the invention relating to the microorganism so that the microorganism can be furnished.
c Microorganisms Excluded from Obligation to be Deposited
(a) Microorganisms which cannot be deposited by a depositary institution designated by the JPO Commissioner for technical reasons or the like
In such a case furnishing of the microorganisms provided in Article 27ter of Regulations under the Patent Act should be guaranteed by the applicant. (Such microorganisms should preferably be deposited with a reliable culture collection.)
(b) Microorganisms easily available for a person skilled in the art stated in "Article 27bis of Regulations under the Patent Act"
More specifically, the following microorganisms are included for example:
(i) Commercially available microorganisms, such as baker's yeast, koji (Aspergillus oryzae), Bacillus natto, etc.
(ii) Microorganisms in a case where it has been evident, prior to filing, that the microorganisms have been stored at a reliable culture collection and are freely furnished from a catalog or the like issued by the culture collection
In this case, the storage number of the microorganism should be stated in the originally attached description.
(iii) Microorganisms which can be produced by a person skilled in the art on the basis of the description.
d Omission of Submission of a Copy of the Receipt of an Original Deposit
If two or more patent applications concerning the same copy of the Receipt of an Original Deposit are filed at the same time, or if filing patent applications concerning a copy of the Receipt of an Original Deposit that has already been submitted, the applicant may omit the submission of the copy of the Receipt of an Original Deposit, according to Article 10(1) and (2) of Regulations under the Patent Act.
For example, the applicant may omit submission of a copy of the Receipt of an Original Deposit under the following cases.
(i) a case of dividing an application
(ii) a case of filing an application claiming internal priority
(iii) a case where the same applicant is filing a second application in which the submission of the same copy of the Receipt of an Original Deposit is necessary
(iv) a case where the applicant is simultaneously filing two or more applications and the submission of the same copy of the Receipt of an Original Deposit is necessary
(v) a case where the applicant is submitting a notice for the change of accession number
(2) Deposit of Genes, Vectors, Recombinant Proteins, Monoclonal Antibodies, Animals and Plants, etc.
If the description is not stated in such a manner that enables a person skilled in the art to produce a gene, a vector, a recombinant protein, a monoclonal antibody, an animal or a plant, etc. in relating to an invention thereof, deposit of them is necessary. In a case of depositing them, a transformant in which a produced gene or vector is introduced (including a transformant producing a recombinant protein), a fused cell (including a hybridoma producing a monoclonal antibody), a fertilized egg, a seed, a plant cell, etc. shall be deposited, and the accession number shall be stated in the originally attached description.
(3) Amendment of Accession Number or the like a An amendment to convert or add an accession number is acceptable because it does not introduce any new technical matter, as long as microbiological or cytobiological characteristics of the biological material are described in the originally attached description, claims or drawings (hereinafter referred to as "originally attached description etc."), to the extent that the biological material can be specified, and the deposit of the biological material can be specified based on the name of the depositary institution, etc. However, if there is doubt about identity of the biological material in changing the accession number, notice may be given of the reason for refusal.
b An amendment converting a storage number of a biological material to an accession number based on the deposit of the biological material with a depositary institution for the purpose of patent procedure, is acceptable because it does not introduce any new technical matter, as long as the biological material used is stored at a reliable culture collection, the storage number of the biological material is explicitly stated in the originally attached description etc., and it is clear that the identity of the biological material is not lost. In such a case, the applicant should make an amendment of the accession number without delay.
c An amendment converting a reference number of biological material to a corresponding accession number is obviously acceptable, as long as the reference number issued by the depositary institution designated by the JPO Commissioner is described in the originally attached description etc. (A reference number corresponds to the number obtained by adding "A" to the head of an accession number in a depositary institution designated by the JPO Commissioner).
d An amendment adding microbiological characteristics of biological material such as a fungus or bacterium is not acceptable because it typically introduces new technical matter unless those characteristics are inherently presented in the originally attached description etc., even if the accession number of the biological material stated in the originally attached description etc. is not changed and microbiological characteristics of the biological material are described in the originally attached description etc. to the extent that the taxonomic species of the biological material such as a fungi and bacterium can be specified. An amendment adding cytological characteristics of an animal or plant cell is treated in the same manner.
(4) Treatment of Patent Applications Claiming Priority
In a case where a claimed invention in an patent application claims priority related to biological material which is not easily available for a person skilled in the art, the invention relating to the biological material can enjoy the effect of the priority provided that the biological material has been deposited with a depositary institution for the purpose of patent procedure or a reliable culture collection, and that the accession number or storage number of the biological material is stated in the description contained in the first application being the basis for priority under the Paris Convention, or in the description contained in the earlier application being the basis for internal priority.
(5) Treatment in a case where an Animal Cell is not deposited in a Depositary Institution due to Mycoplasma Contamination
In principle, an animal cell which is not accepted for deposit due to mycoplasma contamination, is not equivalent to a microorganism excluded from obligation to be deposited (see 1.1.4(1)c).
(Explanation)
In deposit of a microorganism, submission of the microorganism which is not contaminated is originally needed so as to maintain a function and survival capability of the microorganism for working effect of an invention. In a case of a normal microorganism, submission of the microorganism which is not contaminated is possible technically, and when the deposit is not accepted in a depositary institution due to contamination by another microorganism, the microorganism is not treated as an object excluded from obligation to be deposited.
Since, in a case of a cell contaminated with mycoplasma, preventing the contamination is technically possible in general, the cell is not treated as an object excluded from obligation to be deposited even when the deposit is not accepted in a depositary institution, except for a case beyond the control of the depositor.
2. Requirements for Claims
2.1 Clarity Requirement (Article 36(6)(ii))
Determination on clarity requirement relating to biological inventions is carried out according to "Part II Chapter 2 Section 3 Clarity Requirement" in Examination Guidelines. In light of the purpose of Patent Act 36(5), various forms of expression may be used in a claim by the applicants to specify an invention for which a patent is sought.
Therefore, applicants may describe claims using various expression forms so as to specify the biological invention for which a patent is sought. For example, in the case of "an invention of a product", various forms of expression such as operation, function, characteristics, method, use and others may be used to describe matters specifying the invention, in addition to the forms of expression such as a combination of products or a structure of the product.
However, it is to be noted that, in a case where the claim concerning an invention of a product recites a manufacturing process of the product, the statement of the claim(s) meets the requirement of “the invention is clear” only when the invention involves the situation where it is impossible or utterly impractical to define the product by its structure or characteristics at the time of filing.
(1) Invention relating to Nucleic acids and Polypeptides
a Nucleic acids such as Genes
(a) A gene may be described by specifying its nucleotide sequence.
(b) A structural gene may be described by specifying an amino acid sequence of the protein encoded by said gene.
Example: A polynucleotide encoding a protein consisting of an amino acid sequence represented by Met-Asp-... Lys-Glu.
(c) A gene may be described by a combination of the terms such as "deletion substitution or addition" and "hybridize," with functions of the gene in a generic form as follows.
Example 1: A polynucleotide encoding a protein of (i) or (ii) as follows:
(i) a protein whose amino acid sequence is represented by Met-Asp-... Lys-Glu
(ii) a protein derived from the protein of (i) by deletion, substitution or addition of one or more amino acids in the amino acid sequence defined in (i) and having the activity of enzyme A
Example 2: A polynucleotide selected from the group consisting of:
(i) A polynucleotide whose DNA sequence is represented by ATGTATCGG ......
TGCCT
(ii) A polynucleotide which hybridizes under stringent conditions to the polynucleotide whose DNA sequence is complementary to that of the DNA sequence defined in (i), and encodes the protein having the activity of enzyme B
(d) A vector may be described by specifying the complete nucleotide sequence. Further, a vector may be described by specifying each element, the function, or a partial nucleotide sequence of the vector and the function of the partial nucleotide sequence.
(e) A non-coding nucleic acid may be described by specifying the nucleotide sequence. Further, a non-coding nucleotide may be described by specifying the target gene.
Example 1: A probe whose nucleotide sequence is represented by SEQ ID No. X.
Example 2: An siRNA targeting XX gene whose nucleotide sequence is represented by SEQ ID No. X.
b Proteins
(a) A recombinant protein may be described by specifying an amino acid sequence or a nucleotide sequence of a structural gene encoding said amino acid sequence.
Example: A protein consisting of an amino acid sequence represented by Met-Ala-... His-Asp.
(b) A recombinant protein may be described by combining of the terms such as "deletion, substitution or addition" and "X% or more of sequence identity", with functions of the recombinant protein, and if necessary, with origin or source of gene encoding the recombinant protein, in a generic form.
Example 1: A recombinant protein of (i) or (ii) as follows:
(i) a protein whose amino acid sequence is represented by Met-Tyr-... Cys-Leu
(ii) a protein whose amino acid sequence has X% or more of sequence identity of the amino acid sequence of (i) and which has the activity of enzyme A
Example 2: A recombinant protein encoding a polynucleotide of (i) or (ii) as follows:
(i) a polynucleotide whose DNA sequence is represented by ATGTATCGG ...... TGCCT
(ii) a polynucleotide which hybridizes under stringent conditions to the polynucleotide whose DNA sequence is complementary to that of the DNA sequence defined in (i), and encodes the protein having the activity of enzyme B
(c) A protein obtained by isolating from or purifying natural products may be described by specifying the function, physicochemical properties, an amino acid sequence, a production process, etc.
c Antibodies
An antibody may be described by specifying an antigen recognized by the antibody, cross-reactivity, etc. Especially, a monoclonal antibody may be described by specifying an antigen recognized by the monoclonal antibody, a hybridoma which produces the monoclonal antibody, cross-reactivity, an amino acid sequence of CDR in the monoclonal antibody, etc.
Example 1: An antibody to an antigen A.
(Note) An antigen A is necessary to be described by specifying as a substance.
Example 2: An antibody which binds not to an antigen B but to an antigen A.
(Note) An antigen A and an antigen B are necessary to be described by specifying as substances.
Example 3: A monoclonal antibody to an antigen A, produced by a hybridoma in which the accession number is ATCC HB-XXXX.
(Note) An antigen A is necessary to be described by specifying as a substance.
Example 4: A monoclonal antibody in which each amino acid sequence of CDRs 1 to 3 of a heavy chain is represented by SEQ ID No. 1 to 3 and each amino acid sequence of CDRs 1 to 3 of a light chain is represented by SEQ ID No. 4 to 6.
Example 5: A single domain antibody to an antigen A.
(Note) An antigen A is necessary to be described by specifying as a substance.
(2) Invention relating to Microorganisms, Animals and Plants
a Fused Cells
A fused cell may be described by specifying parent cells of the fused cell, a typical gene or membrane protein of the fused cell, the characteristics of the fused cell, a production process of the fused cell, etc.
If the fused cell is deposited, the fused cell may be described by specifying the accession number.
Example 1: A fused cell in which a mouse spleen cell sensitized with an antigen A is fused to a myeloma cell, and which has capability of producing an antibody to the antigen A.
(Note) An antigen A is necessary to be described by specifying as a substance.
Example 2: A hybridoma in which the accession number is FERM P-XXXXX, and which produces an anti-XX monoclonal antibody.
b Dedifferentiated Cells
A dedifferentiated cell may be described by specifying factors contributing to dedifferentiation which are introduced to the differentiated cell (reprogramming factors), a production process of the dedifferentiated cell, etc.
Example: An induced pluripotent stem cell which is produced by introducing A factor, B factor, and C factor.
c Transformants
A transformant may be described by specifying a gene (or vector) to be introduced, etc.
Example 1: A transformant which is transformed by a vector comprising a gene encoding a protein whose amino acid sequence is represented by Met-Asp- ... Lys-Glu.
Example 2: A plant which is transformed by a toxin gene having a nucleotide sequence of ATGACT...... and in which the toxic gene is expressed.
Example 3: A transgenic non-human mammal, having a recombinant DNA obtained by linking a structural gene encoding any protein to the regulatory region of a gene encoding a milk protein of casein, and secreting said protein into milk.
d Microorganisms (obtained by means other than genetic engineering)
A microorganism may be specified by description in a claim by combining of a name of the microorganism, a typical gene and characteristics of the microorganism, a production process of the microorganism, etc.
If the microorganism is deposited, the microorganism may be specified by the accession number.
Example 1: A mesenchymal stem cell which is isolated from human marrow, expresses cell-surface antigens A, B, C, D, and E, and does not express cell-surface antigens X, Y, and Z.
Example 2: Bacillus subtilis FERM P-XXXXX strain.
Example 3: A murine tumor cell line in which the accession number is FERM P-XXXXX.
e Animals and Plants (obtained by means other than genetic engineering)
An animal or a plant may be specified in a claim by combining a name of the animal or plant, a typical gene and characteristics of the animal or plant, a production process of the animal or plant, etc.
If the animal or plant is deposited, the animal or plant may be specified by the accession number.
Example 1: A rice plant obtained by treating a rice plant in a growth process with a composition comprising a phytohormone X.
Example 2: A plant belonging to Castanea crenata (Japanese chestnut) having the accession number of ATCC-XXXXX whose bark contains catechol tannin and pyrogallol tannin in the ratio of (X 1 to X 2 ): (Y 1 to Y 2 ) and has the content of catechol tannin of Z 1 to Z 2 ppm (weight ratio), or its mutant having said characteristics.
Example 3: A watermelon obtained by crossing a diploid watermelon with a tetraploid watermelon obtained by polyploidizing a diploid watermelon, whose somatic cell has 33 chromosomes.
Example 4: A process for producing a cabbage which is obtained by crossing a cabbage strain having the accession number of ATCC-XXXXX as a seed parent with another cabbage as a pollen parent, and has resistance for a herbicide X.
3. Drawings
When photographs are attached as drawings, black-and-white photographs should be used. Color photographs may be submitted as references.
4. Sequence Listing
(1) If a nucleic acid sequence consisting of 10 or more nucleotides, or an amino acid sequence of a protein or peptide consisting of 4 or more L-amino acids is described in a description, claims or drawings, a "Sequence Listing" including the sequence prepared by using code data in accordance with "Guidelines for the preparation of description which contain nucleotide and/or amino acid sequence" (see JPO website) should be described at the end of the description as a part of it (see note 17 of Form 29, Article 24 of Regulations under the Patent Act).
(2) If a nucleotide sequence or an amino acid sequence is described in the claims, the sequence described in the "Sequence Listing" prepared in accordance with "Guidelines for the preparation of description which contain nucleotide and/or amino acid sequence" may be cited.
(3) Properties of the sequence may be described as free text in a sequence listing. At that time, the content of free text may add a caption of "sequence listing free text" and should be described in the description.
5. Requirements for Patentability
5.1 Eligibility for Patent and Industrial Applicability (the main paragraph in Article 29(1))
Determination on eligibility for patent and industrial applicability relating to biological inventions is carried out according to “Part III, Chapter 1, Eligibiity for Patent and Industrial Applicability” in Examination Guidelines.
For example, determination on eligibility for patent and industrial applicability is carried out as follows.
(1) Matter Not Eligible for “Patent”
A mere discovery which is not a creation is not considered as a statutory "invention".
Example: A merely discovered organism existing in nature
However, a microorganism which is artificially isolated from natural surroundings has creativity.
(2) Invention Not Eligible for "Industrial Applicability"
In an invention relating to biological material, if its applicability is not sated in the description, the claims, or drawings, and the applicability cannot be inferred, the invention is considered to be a commercially inapplicable invention. Therefore, the invention is not eligible for an "industrial applicable".
5.2 Novelty (Article 29(1)(i) to (iii))
Determination on novelty relating to biological inventions is carried out according to “Part III, Chapter 2, Novelty and Inventive Step” in Examination Guidelines.
For example, it is determined as follows.
(1) Invention relating to Nucleic acids and Polypeptides
a Proteins
If a protein as an isolated and purified single substance is publicly known, and a claimed invention relating to a recombinant protein specified by a process of production is not distinguished from the publicly known protein as a product, said recombinant protein does not have novelty.
However, if a recombinant protein is obtained which is different from the publicly known protein in its glycan or the like, by using the different microorganisms, animals or plants, even though the recombinant protein has the same amino acid sequence as the publicly known protein, an invention relating to the recombinant protein described by specifying a process of production has novelty.
b Antibodies
(a) If an antigen A has novelty, an invention of an antibody to the antigen A has novelty in general. However, if a monoclonal antibody to publicly known antigen A' is publicly known and the antigen A has the same epitope as that of antigen A' because the antigen A is partially modified from the publicly known antigen A', a monoclonal antibody to the antigen A' also binds to the antigen A. In such a case, an invention of "a monoclonal antibody to the antigen A" cannot be distinguished from the publicly known monoclonal antibody as a product. Therefore, the invention does not have novelty.
(b) An invention relating to an antibody specified by cross-reactivity to an antigen B different from the antigen A, such as "an antibody which binds not to an antigen B but to an antigen A" is not considered that the cross-reactivity represents a definition of the specific product, if an antibody to the antigen A is publicly known and there is no particular technical significance to specify the antibody described by the cross-reactivity (e.g. when it is evident that the antibody to the publicly known antigen A does not bind to the antigen B because the antigen B has no similarity to the antigen A in terms of function, structure, etc.). Therefore, the invention does not have novelty, since the invention cannot be distinguished from the publicly known antibody as a product in general.
(2) Invention relating to Microorganisms, Animals and Plants
a Differentiated Cells
Even if a stem cell itself has novelty or a process of inducing differentiation has novelty, in a case where a cell obtained by performing differentiation induction of the stem cell cannot be distinguished from the publicly known differentiated cells as a product (for example, in a case where the obtained cell expresses only a publicly known differentiation marker), an invention of the obtained cell does not have novelty.
5.3 Inventive Step (Article 29(2))
Determination on inventive step relating to biological inventions is carried out according to “Part III, Chapter 2, Novelty and Inventive Step” in Examination Guidelines.
For example, it is determined as follows.
(1) Invention relating to Nucleic acids and Polypeptides a Nucleic acids such as Genes
(a) If a protein A has novelty and an inventive step, an invention of a gene encoding the protein A involves an inventive step.
(b) If a protein A is publicly known but the amino acid sequence is not publicly known, an invention of a gene encoding the protein A does not involve an inventive step when a person skilled in the art could determine the amino acid sequence of the protein A easily at the time of filing. However, if the gene is described by a specific nucleotide sequence and has advantageous effects that a person skilled in the art cannot expect in comparison with other genes having a different nucleotide sequence encoding the protein A, the invention of said gene involves an inventive step.
(c) If an amino acid sequence of a protein A is publicly known, an invention of a gene encoding the protein A does not involve an inventive step. However, if the gene is described by a specific nucleotide sequence and has advantageous effects that a person skilled in the art cannot expect in comparison with other genes having a different nucleotide sequence encoding the protein A, the invention of said gene involves an inventive step.
(d) If a structural gene is publicly known, an invention of a structural gene which has high sequence identity to the publicly known structural gene and has the same property and function as that of the publicly known structural gene, does not involve an inventive step. However, if the claimed structural gene has advantageous effects that a person skilled in the art cannot expect in comparison with the publicly known structural gene, the invention of said structural gene involves an inventive step.
(e) If a structural gene and the conserved motif are publicly known, an invention of a structural gene which has the same property and function as that of the publicly known structural gene and has the conserved motif, does not involve an inventive step. However, if the claimed structural gene has advantageous effects that a person skilled in the art cannot expect in comparison with the publicly known structural gene, the invention of said structural gene involves an inventive step.
(f) If a structural gene is publicly known, an invention of a promoter of a structural gene cluster including a structural gene which has high sequence identity to said structural gene and has the same property and function as that of said structural gene, does not involve an inventive step. However, if the claimed promoter has advantageous effects that a person skilled in the art cannot expect, the invention of said promoter involves an inventive step.
(g) If both of a vector and a gene to be introduced are publicly known, an invention of a recombinant vector obtained by a combination of them does not involve an inventive step. However, if the recombinant vector obtained by specifying a combination of them has advantageous effects that a person skilled in the art cannot expect, the invention of said recombinant vector involves an inventive step.
(h) If an invention of a gene A does not have novelty or involve an inventive step, an invention of a primer or a probe for detecting the gene A does not involve an inventive step. However, if an invention of the primer or probe which is further specified by a nucleotide sequence, and the specified primer or probe has advantageous effects that a person skilled in the art cannot expect, the invention of said primer or probe involves an inventive step.
(i) In a case where a nucleotide sequence of a gene A is publicly known, if it is not difficult to select a target domain, an invention of an antisense nucleic acid or siRNA to the gene A does not involve an inventive step. However, if the antisense nucleic acid or siRNA has advantageous effects that a person skilled in the art cannot expect, the invention of said antisense nucleic acid or siRNA involves an inventive step.
(j) An invention of an SNP or mRNA expression profile found by using a publicly known analysis technique which is used to statistically and comprehensively find marker candidates for various diseases to identify a marker for a specific disease, does not involve an inventive step. However, if it is difficult to apply the analysis technique to the specific disease since the involvement of genetic factors relating to said disease has been denied, or if an odds ratio, sensitivity or specificity relating to the identified SNP or mRNA expression profile is indicated and the odds ratio, sensitivity or specificity has advantageous effects that a person skilled in the art cannot expect, the invention of said SNP or mRNA expression profile involves an inventive step.
b Proteins
If a protein is publicly known, an invention of a mutant of the protein which has the same property and function as that of the protein, does not involve an inventive step. However, if the claimed mutant of the protein has advantageous effects that a person skilled in the art cannot expect in comparison with the publicly known protein, the invention of said mutant of the protein involves an inventive step.
c Antibodies
If an antigen A is publicly known and it is evident that the antigen A has immunogenicity (for example, the antigen A is a polypeptide with a large molecular weight), an invention of "an antibody to the antigen A" does not involve an inventive step. However, if the invention is further specified by other characteristics and has advantageous effects that a person skilled in the art cannot expect, the invention of said antibody involves an inventive step.
(2) Invention relating to Microorganisms, Animals and Plants
a Fused Cells
If both of parent cells are publicly known, an invention of a fused cell obtained by fusing parent cells using a means which a person skilled in the art commonly uses does not involve an inventive step. However, if the fused cell obtained by a specific combination of them has advantageous effects that a person skilled in the art cannot expect, the invention of said fused cell involves an inventive step.
b Transformants
(a) If both of a host and a gene to be introduced are publicly known, an invention of a transformant obtained by using a means which a person skilled in the art commonly uses does not involve an inventive step. However, if the transformant obtained by a specific combination of them has advantageous effects that a person skilled in the art cannot expect, the invention of said transformant involves an inventive step.
(b) If an animal or a plant before genetic recombination and the introduced or deleted gene are publicly known, an invention of an animal or a plant recombined by the gene transfer method or the gene deletion method, which a person skilled in the art commonly uses, does not involve an inventive step. However, if it is difficult to introduce or delete the gene to/from the animal or plant before gene recombination, or if the characteristics of the recombined animal or plant have advantageous effects in comparison with characteristics which are expected in as case where the gene is introduced or deleted to/from the animal or plant before gene recombination, the invention of said animal or plant involves an inventive step.
c Microorganisms (obtained by means other than genetic engineering)
(a) An invention of a microorganism obtained by performing mutating treatment of a publicly known species, which a person skilled in the art commonly uses, does not involve an inventive step. However, if the microorganism has advantageous effects that a person skilled in the art cannot expect, the invention of said microorganism involves an inventive step.
(b) In a case of fungi or bacteria, a person skilled in the art usually and easily ascertains the applicability (for example, material productivity) and effects of publicly known species within classification hierarchy (for example, "genus") for which it is known that they have the same property, by culturing each microorganism. Therefore, an invention relating to the use of a fungus or bacterium does not involve an inventive step in general, if the fungus or bacterium used in the invention is a taxonomically known species and belongs to the same classification hierarchy (for example, "genus") as another fungus or bacterium for which the same mode of use as the invention is known, and it is publicly known that the fungus or bacterium belonging to the same classification hierarchy has the same property. However, the invention relating to the use of the fungus or bacterium has advantageous effects that a person skilled in the art cannot expect, the invention involves an inventive step.
d Animals and Plants (obtained by means other than genetic engineering)
An invention of an animal or a plant obtained by using a means which a person skilled in the art commonly uses does not involve an inventive step. However, if the animal or plant has advantageous effects that a person skilled in the art cannot expect, the invention of said animal or plant involves an inventive step.
6. Cases
This chapter explains the practice of examination on biological inventions, on the basis of specific cases.
(Points to note)
These cases are prepared for the purpose of explaining the application of examination on biological inventions. Therefore, it is to be noted that the descriptions of the claims, etc. in these cases are modified, e.g., simplified to make the explanation of biological inventions easier to understand. Additionally, it is to be noted that it does not mean that there is no reason for refusal other than reasons considered in each case.
(1) Cases relating to Requirements of Unity of Invention, Description Requirements and Requirements for Patentability
List of Cases
(○ means the requirement which is considered in the case.)
(2) Cases relating to Determination of Necessity for Deposit of Microorganisms, etc.
This section explains about the determination whether or not microorganisms, etc. ("microorganisms, etc." here include microorganisms, plants and animals) are required to be deposited prior to filing the application, on the basis of specific cases.
For information on general matters relating to the determination of necessity for deposit, see "1.1.4 Deposit and Furnishing of Biological Material".
(Points to note)
In cases relating to determination of necessity for deposit of microorganisms, etc., each case shall not be taken to mean that there is no reason for refusal such as lack of novelty/inventive step.
6.1 Cases relating to Requirements of Unity of Invention, Description Requirements and Requirements for Patentability
[Case 1] Case relating to Unity of InventionTitle of the Invention
Polynucleotides
Claims
[Claim 1] An isolated polynucleotide selected from DNA sequences represented by SEQ ID NOs: 1-10.
Overview of the description
This invention relates to the cDNAs of 1000-2000 bp obtained from the human liver cDNA library. It is found that these polynucleotides encode a protein having serine protease activity despite having a different structure from each other. These polynucleotides have low sequence identity with each other.
The polynucleotides according to Claim 1 have a technical feature that the polynucleotides encode proteins having serine protease activity, but the technical feature is not a special technical feature because the technical feature is publicly known from the prior art.
The polynucleotides according to Claim 1 would be regarded as having the same or corresponding special technical feature if the polynucleotides have a common property or activity in all the alternatives, and shared a significant structural element that is essential to the common property or activity. In this case, all polynucleotides represented by SEQ ID NOs: 1-10 have a common property of encoding proteins having serine protease activity, on the other hand, these polynucleotides have low sequence identity with each other and do not have a common structure, namely a significant structural element. The sugar-phosphate skeleton cannot be considered as a significant structural element since it is shared by all polynucleotide molecules.
Therefore, 10 polynucleotides according to Claim 1 do not have the same or corresponding technical feature.
[Case 2] Case relating to Unity of Invention
Title of the Invention
Polynucleotides
Claims
[Claim 1] An isolated polynucleotide selected from DNA sequences represented by SEQ
ID NOs: 1-10.
Overview of the description
This invention relates to the cDNAs of 400-500 bp obtained from the human liver cDNA library. The polynucleotides of the invention claimed in Claim 1 all share a common significant structural element and their corresponding mRNAs are expressed only in the hepatocytes of patients with disease Y. The corresponding mRNAs are not expressed in the hepatocytes of healthy individuals.
(Supplemental explanation)
The polynucleotides according to Claim 1 would be determined as having the same or corresponding technical feature if the polynucleotides had a common property or activity, and shared a significant structural element that is essential to the common property or activity in all the alternatives.
In this case, the description discloses that the polynucleotides represented by SEQ ID NOs: 1-10 share a common property, that is, the polynucleotides correspond to mRNAs expressed only in patients with disease Y. Moreover, the polynucleotides represented by SEQ ID NOs: 1-10 share a significant structural element that is essential to the common property, i.e., a common significant structural element capable of detecting the mRNA of patients with disease Y. Since both of these requirements are met, the polynucleotides of the invention claimed in Claim 1 have the same or corresponding special technical feature with each other.
[Case 3] Case relating to Unity of Invention
Title of the Invention
Fusion proteins capable of inducing antibodies to Escherichia coli
Claims
[Claim 1] A fusion protein comprising carrier protein X bound to a polypeptide consisting of an amino acid sequence represented by SEQ ID NO: 1, 2 or 3.
Overview of the description
Carrier protein X consisting of 1000 amino acid residues, and has a function to improve the stability of the fusion protein in the blood stream. The polypeptide consisting of the amino acid sequence represented by SEQ ID NO: 1, 2 or 3 is a small immunogenic epitope isolated from different antigenic regions of E. coli, which has a length of 10-20 residues, and it is confirmed that the fusion protein bound to carrier protein X induces an antibody specific to E. coli. However, the amino acid sequences represented by SEQ ID NOs: 1, 2 and 3 do not share any significant structural element with each other.
The fusion protein according to Claim 1 has a technical feature that the fusion protein comprises carrier protein X, but the technical feature is not a special technical feature because the technical feature is publicly known from the prior art.
The fusion protein according to Claim 1 would be determined as having the same or corresponding technical feature if the fusion protein had a common property or activity, and shared a significant structural element that is essential to the common property or activity in all the alternatives. However, in this case, the common structure shared in the fusion protein is only carrier protein X. While the fusion proteins have the common property that the fusion protein induces an antibody specific to E. coli, the common property cannot be exhibited only by immunizing with single carrier protein X, and the polypeptide represented by SEQ ID NO: 1, 2 or 3 is required to exert this common property. In this case, the fact that three fusion proteins have the common property is not sufficient to have the same or corresponding special technical feature.
This is because the polypeptide represented by SEQ ID NO: 1, 2 or 3 imparting the common property does not share a significant structural element, the common structure of carrier protein X does not impart the common property to induce an antibody specific to E. coli, and the fusion protein inducing antigenic response specific to E. coli is known in the prior art.
Therefore, the fusion protein according to Claim 1 does not have the same or corresponding special technical feature.
[Case 4] Case relating to Unity of Invention
Title of the Invention
Nucleic acid molecules encoding dehydrogenase A
Claims
[Claim 1] An isolated nucleic acid molecule consisting of a nucleotide sequence selected from SEQ ID NO: 1, 2 or 3.
Overview of the description
Three nucleic acid molecules encoding dehydrogenase A comprise a conserved motif sequence defining a function of dehydrogenase A. These three nucleic acid molecules are isolated from three different sources (mouse, rat and human). These three nucleic acid molecules have high sequence identity (85-95% of sequence identity) as a whole at both the nucleic acid sequence and amino acid sequence levels.
The nucleic acid molecule according to Claim 1 has a technical feature that the nucleic acid molecule encodes dehydrogenase A, but the technical feature is not a special technical feature because the technical feature is publicly known from the prior art.
The nucleic acid molecules of the invention claimed in Claim 1 would be determined as having the same or corresponding technical feature if the nucleic acid molecules had a common property or activity, and shared a significant structural element that is essential to the common property or activity in all the alternatives.
However, in this case, the nucleic acid molecule which encodes dehydrogenase A and has the common structural element has already been isolated from a different source (monkey), and the molecule has been publicly known. Since the functional and structural similarity between the nucleic acid molecules according to Claim 1 does not make any contribution over the prior art, the similarity is not a special technical feature.
Therefore, the nucleic acid molecules of the invention claimed in Claim 1 do not have the same or corresponding special technical feature.
[Case 5] Case relating to Unity of Invention
Title of the Invention
Method of screening and compounds identified by the same
Claims
[Claim 1] A method for identifying compounds that are antagonists of receptor R comprising the steps of: step 1: contacting cells expressing receptor R on their outer membrane with its natural ligand; step 2: contacting the cells bound to the ligand with a candidate compound selected from a library of compounds; and step 3: observing any change in the binding state of the natural ligand.
[Claim 2] A compound X having Chemical formula 1.
[Claim 3] A compound Y having Chemical formula 2.
[Claim 4] A compound Z having Chemical formula 3.
Overview of the description
Receptor R and its natural ligand are recognized as a drug target. Compounds that act as antagonists of receptor R have been proposed to have physiological effects that may be useful in therapeutic treatment. The aim of the invention is to identify lead compounds as a basis for further screening and testing of combinatorial libraries.
A library is used as providing structurally different compounds with high possibility.
The method of the invention claimed in Claim 1 is useful to identify compounds affecting the physiological effect of binding of the natural ligand to receptor R.
Actually, compounds X, Y and Z are identified as compounds having such an effect, but these compounds do not share a significant structural element. The relationship between the structures of the compounds of the inventions claimed in Claims 2-4 and the antagonistic function is unknown, as is the relationship between the antagonistic function of compounds and the structure of receptor R.
A special technical feature of the method of the invention claimed in Claim 1 is the step of observing the effect of the candidate compounds on ligand binding in a screening assay. Neither the same nor a corresponding special technical feature is present in any of compounds X, Y and Z of the inventions claimed in Claims 2-4.
The screening method of the invention claimed in Claim 1 is neither the method of preparing nor a method of using the compounds X, Y and Z of the inventions claimed in Claims 2-4. In the absence of any suggestion of the specific structure required for a compound to act as an antagonist of receptor R, there is no single general inventive concept that links the screening method of the invention claimed in Claim 1 to the compounds of the inventions claimed in Claims 2-4. Therefore, the compounds do not have the same or corresponding special technical feature.
Considering unity of invention relating to the compounds of the inventions claimed in Claims 2-4, the compounds X, Y and Z would be determined as having the same or corresponding technical feature if the compounds have a common property or activity, and shared a significant structural element that is essential to the common property or activity in all the alternatives.
While the compounds X, Y and Z have the common property that the compounds function as an antagonist of receptor R, there is no suggestion of a common significant structural element, and hence, there is no disclosure of the same or corresponding special technical feature. Therefore, the compounds of the inventions claimed in Claims 2-4 do not have a same or corresponding special technical feature.
[Case 6] Case relating to Unity of Invention
Title of the Invention
Interleukin-1 and DNA encoding the same
Claims
[Claim 1] An isolated interleukin-1 having the amino acid sequence represented by SEQ ID NO: 1.
[Claim 2] An isolated DNA molecule encoding the interleukin-1 according to Claim 1.
Overview of the description
This invention relates to interleukin-1 which is water-soluble cytokine related to lymphocytic activation. Interleukin-1 is isolated and purified for the first time by this invention. SEQ ID NO: 1 represents an amino acid sequence of the interleukin-1, and SEQ ID NO: 2 represents the nucleotide sequence of a DNA molecule encoding the interleukin-1.
(Supplemental explanation)
Since the DNA molecule according to Claim 2 encodes interleukin-1 making the contribution over the prior art, interleukin-1 and DNA encoding the interleukin-1 have the corresponding special technical feature. Therefore, the inventions claimed in Claims 1 and 2 have the same or a corresponding special technical feature.
[Case 7] Case relating to Enablement Requirement
Title of the Invention
Full-length cDNA
Claims
[Claim 1] A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 5.
Overview of the description
A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 5 is cDNA of 3000 bp obtained from the human liver cDNA library, and encodes a polypeptide consisting of 1000 amino acid residues represented by SEQ ID NO: 6.
As a result of homology search of the DNA sequence represented by SEQ ID NO: 5 and the amino acid sequence represented by SEQ ID NO: 6 using DNA and amino acid sequence databases published before filing the application, there is found no other DNA and amino acid sequence having 30% or more sequence identity to the DNA represented by SEQ ID NO: 5 and the amino acid sequence represented by SEQ ID NO: 6. On the other hand, the amino acid sequence represented by SEQ ID NO: 6 is proved to have a potential site of glycosylation in the polypeptide.
Therefore, the polynucleotide of the invention claimed in Claim 1 is assumed to encode a new glycoprotein, whose specific function is unknown, that may be used for developing a new drug.
In regard to an invention of a product, the description must be stated in such a manner so as to enable a person skilled in the art to produce and use the product.
Since there are various kinds of function of glycoprotein, even though the polynucleotide encodes a glycoprotein, a specific function of the glycoprotein is unknown.
While the fact that proteins having high sequence identity probably have similar functions to each other was the common general knowledge at the time of filing, there was no publicly known protein, prior to filing the application, having high sequence identity with the polypeptide encoded by the polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 5.
Then, the specific function of the protein encoded by the polynucleotide cannot be expected.
Since the specific function of the polynucleotide is unknown, how to use the polynucleotide is also unknown.
Therefore, the description cannot be regarded as stating the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention claimed in Claim 1. [Measures of the applicant] Generally, the reason for refusal stated above shall not be overcome.
(Supplemental explanation)
The "specific function" stated here means a "function from which a specific use with technical meanings can be assumed."
[Case 8] Case relating to Enablement Requirement
Title of the Invention
Full-length cDNA
Claims
[Claim 1] A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 7.
Overview of the description
A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 7 is cDNA of 2400 bp obtained from the human liver cDNA library, and encodes a polypeptide consisting of 800 amino acid residues represented by SEQ ID NO: 8.
As a result of homology search of the DNA sequence represented by SEQ ID NO: 7 and the amino acid sequence represented by SEQ ID NO: 8 using DNA and amino acid sequence databases published prior to filing the application, the DNA sequence and amino acid sequence have 20-30% of sequence identity to the DNA sequence encoding factor WW1 of mammals such as rat and the amino acid sequence of the factor WW1 described in document A, B etc., respectively.
Therefore, the polynucleotide of the invention claimed in Claim 1 is assumed to encode human factor WW1 and to be useful.
In regard to an invention of a product, the description must be stated in such a manner so as to enable a person skilled in the art to produce and use the product.
The grounds that the polynucleotide encodes human factor WW1 is only based on the fact that the DNA sequence and amino acid sequence have 20-30% of sequence identity to the DNA sequence encoding factor WW1 of mammals such as rat and to the amino acid sequence of the factor WW1.
In general, in a case where two polynucleotides (polypeptides) have 20-30% of sequence identity with each other, they probably do not have the same specific function.
Further, there is no common general knowledge at the time of filing that the polynucleotide probably encodes factor WW1 when a polynucleotide (polypeptide) has 20-30 % of sequence identity to a polynucleotide (factor WW1) encoding factor WW1.
Therefore, since the polynucleotide of the invention claimed in Claim 1 probably do not actually encode factor WW1, a specific function of the polynucleotide is unknown. Since the specific function of a protein encoded by the polynucleotide cannot be expected, how to use the polynucleotide is also unknown.
Accordingly, the description cannot be regarded as stating the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention claimed in Claim 1. [Measures of the applicant] The reason for refusal stated above may be overcome if the applicant proves in a written opinion that the polynucleotide of the invention claimed in Claim 1 encodes "human factor WW1" with the certification of the function of the actually expressed protein and a logical explanation.
(Supplemental explanation)
In a case where the above logical explanation is based on the knowledge of the publicly known conserved region prior to filing the application, and when it is determined that a person skilled in the art could be easily obtain a polynucleotide encoding "factor WW1" by a PCR method, etc. using a DNA primer prepared on the basis of a DNA sequence of the conserved region and the polynucleotide does not have unexpected advantageous effect, the invention claimed in Claim 1 lacks inventive step.
The "specific function" stated here means a "function from which a specific use with technical meanings can be assumed."
The "specific function" of factor WW1, i.e. the "function from which a specific use with technical meaning can be assumed" is known.
[Case 9] Case relating to Enablement Requirements
Title of the Invention
Full-length cDNA
Claims
[Claim 1] A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 9.
Overview of the description
A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 9 is cDNA of 2400 bp obtained from the human liver cDNA library, and encodes a polypeptide consisting of 800 amino acid residues represented by SEQ ID NO: 10.
As a result of homology search of the DNA sequence represented by SEQ ID NO: 9 and the amino acid sequence represented by SEQ ID NO: 10 using DNA and amino acid sequence databases published prior to filing the application, the DNA sequence and amino acid sequence have 20-30% of sequence identity to the DNA sequence encoding rat factor ZZ1 and the amino acid sequence of the rat factor ZZ1 described in document A, the DNA sequence encoding pig factor ZZ2 and the amino acid sequence of the pig factor ZZ2 described in document B, and the DNA sequence encoding an antagonist of monkey factor ZZ1 receptor and the amino acid sequence of the antagonist of monkey factor ZZ1 receptor described in document C, respectively.
Therefore, the polypeptide of the invention claimed in Claim 1 encodes a protein related to human factor ZZ, and may be used to treat diseases related to factor ZZ.
In regard to an invention of a product, the description must be stated in such a manner so as to enable a person skilled in the art to produce and use the product.
Since the proteins related to human factor ZZ comprises factor ZZ1, factor ZZ2 and the antagonist of factor ZZ1 receptor which have different functions from each other, a specific function of the protein related to human factor ZZ is still unknown in the description even though the polynucleotide of the invention claimed in Claim 1 encodes the protein related to human factor ZZ.
While the fact that proteins having high sequence identity probably have similar functions with each other was the common general knowledge at the time of filing, there was no publicly known protein, prior to filing the application, having high sequence identity with a polypeptide encoding the polynucleotide of the invention claimed in Claim 1.
Therefore, the specific function of a protein encoded by the polynucleotide cannot be expected.
Since the specific function of the polynucleotide is unknown, how to use the polynucleotide is also unknown.
Accordingly, the description cannot be regarded as stating the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention claimed in Claim 1. [Measures of the applicant] Even if it proved in written opinion that the polynucleotide of the invention claimed in Claim 1 encodes "human factor ZZ1" of proteins related to ZZ factor, the reason for refusal stated above shall not be overcome in general.
(Supplemental explanation)
Even though the description states that “the DNA sequence and amino acid sequence have 20-30% of sequence identity to the DNA sequence encoding rat factor ZZ1 and the amino acid sequence of the rat factor ZZ1, the DNA sequence encoding pig factor ZZ2 and the amino acid sequence of the pig factor ZZ2, and the DNA sequence encoding an antagonist of monkey factor ZZ1 receptor and the amino acid sequence of the antagonist of monkey factor ZZ1 receptor, respectively”, and that “the polypeptide encodes a protein related to human factor ZZ”, it cannot be assumed directly that “the polynucleotide encodes human factor ZZ1” on the basis of the common general knowledge at the time of filing.
The "specific function" stated here means a "function from which a specific use with technical meanings can be assumed."
Factor ZZ1, factor ZZ2 and the antagonist of factor ZZ1 receptor which are proteins related to factor ZZ are known to have different "specific functions" each other, i.e. "functions from which a specific use with technical meanings can be assumed."
[Case 10] Case relating to Inventive Step
Title of the Invention
Full-length cDNA
Claims
[Claim 1] A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 11.
Overview of the description
A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 11 is cDNA of 2700 bp obtained from the human liver cDNA library, and encodes a polypeptide consisting of an amino acid sequence of 900 amino acids represented by SEQ ID NO: 12.
As a result of homology search of DNA sequence represented by SEQ ID NO: 11 and the amino acid sequence represented by SEQ ID NO: 12 using DNA and amino acid sequence databases published prior to filing the application, the DNA sequence has 80% of sequence identity to the DNA sequence encoding rat factor XX1 described in document A and the amino acid sequence has 85% of sequence identity to the amino acid sequence of rat factor XX1 described in document A.
Therefore, the polynucleotide of the invention claimed in Claim 1 is assumed to encode human factor XX1 and to be useful.
It was a well-known problem to obtain a polynucleotide encoding one protein prior to filing the application.
On the basis of the common general knowledge that a polynucleotide encoding one mammal protein and a polynucleotide encoding a homolog protein from another mammal have high sequence identity to each other in general, it was a well-known technique to obtain the polynucleotide encoding the homolog protein by PCR method, etc. using a part of the polynucleotide encoding the known mammal protein as a PCR primer.
Therefore, a person skilled in the art would easily conceive the idea of using a
PCR primer prepared on the basis of the DNA sequence of the polynucleotide encoding rat factor XX1 described in document A, and obtaining the polynucleotide encoding human factor XX1 from a human cDNA library so as to obtain human factor XX1. It is also considered that the polynucleotide of the invention claimed in Claim 1 does not have any advantageous effect which cannot be expected from the document A and well-known art. [Measures of the applicant] The reason for refusal stated above may be overcome if the applicant proves in a written opinion that there was specific difficulty to obtain the polynucleotide of the invention claimed in Claim 1 in view of the state of the art at the time of filing.
(Supplemental explanation)
The specific function of factor XX1, i.e., the function from which a specific use with technical meanings can be assumed, is known.
[Case 11] Case relating to Inventive Step and Enablement Requirement
Title of the Invention
Full-length cDNA
Claims
[Claim 1] A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 17.
Overview of the description
A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 17 is cDNA of 2700 bp obtained from the human liver cDNA library, and encodes a polypeptide consisting of an amino acid sequence of 900 amino acids represented by SEQ ID NO: 18.
The polynucleotide is actually expressed, and it is found that the polynucleotide has activity of enzyme X.
[Case 12] Case relating to Inventive Step and Enablement Requirement
Title of the Invention
DNA fragments
Claims
[Claim 1] A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 13.
Overview of the description
A cDNA library is constructed from human liver using oligo (dT) primers.
The DNA sequence represented by SEQ ID NO: 13 is one of the sequences having a length of 500 bp which are analyzed using a sequencer.
The polynucleotide consisting of the nucleotide sequence represented by SEQ ID NO: 13 is a part of a structural gene, and it can be used as a probe in one of the steps to obtain the full-length DNA.
However, there is no example indicating that the full-length DNA was actually obtained, and there is no disclosure of the function or biological activity of the polynucleotide and its corresponding protein.
It is a well-known art to construct cDNA libraries from human organs such as the liver, and to analyze the sequence of cDNA randomly chosen from the library using a sequencer.
Therefore, a person skilled in the art would easily construct cDNA libraries and analyze the sequence of cDNA randomly chosen from the library using a sequencer so as to obtain sequence information by applying the well-known art. Nor is it found that the polynucleotide of the invention claimed in Claim 1 have an unexpected advantageous effect on the basis of the well-known art.
Article 36(4)(i) (Enablement Requirement)
In regard to an invention of a product, the description must be stated in such a manner so as to enable a person skilled in the art to produce and use the product.
It is stated in the description that the polynucleotide of the invention claimed in Claim 1 can be used as a probe in one of the steps to obtain a full-length DNA.
However, there is no description on a function or biological activity of the protein encoded by the corresponding full-length DNA. Moreover, the function or biological activity cannot be expected.
The use of a DNA fragment to obtain the full-length DNA, whose corresponding protein's function and biological activity are unknown, is not considered to "enable a person skilled in the art to use the product". Therefore, the description cannot be regarded as stating the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention claimed in Claim 1.
[Case 13] Case relating to Inventive Step and Enablement Requirement
Title of the Invention
DNA fragments
Claims
[Claim 1] A polynucleotide consisting of the DNA sequence represented by SEQ ID NO: 16.
Overview of the description
The polynucleotide is one of cDNAs having the length of 500 bp which is found in a cDNA library constructed from the hepatocytes of patients with disease Y, but not found in those of healthy individuals.
It is confirmed by northern hybridization that the corresponding mRNAs are expressed only in the hepatocytes of patients with disease Y. Therefore, the polynucleotide can be used to diagnose disease Y.
Moreover, DNA sequence and amino acid sequence having 30% or more sequence identity to the DNA sequence represented by SEQ ID NO: 16 were not found. [Overview of the reason for refusal] None
(Supplemental explanation)
The polynucleotide of the invention claimed in Claim 1 has advantageous effect that the polynucleotide can be used for diagnosing disease Y and this effect cannot be expected from the prior art.
[Case 14] Case relating to Inventive Step and Enablement Requirement
Title of the invention
SNP
Claims
[Claim 1] A polynucleotide having consecutive 20-100 bases including the 100 th base (polymorphic site) of the DNA sequence set forth in SEQ ID NO: 14.
Overview of the description
Sequencing and comparison of sequences of 500 bases in the XX locus in genomic DNA from 10 people revealed that 2 people had DNA having the sequence set forth in SEQ ID NO: 14 and 8 people had DNA having the sequence set forth in SEQ ID NO: 15. The DNA having the sequence set forth in SEQ ID NO: 15 is publicly known prior to the filing of the application. The two sequences are only different in that the 100 th base in the DNA sequence set forth in SEQ ID NO: 15 is c, while the 100 th base in the DNA sequence set forth in SEQ ID NO: 14 is g.
A polynucleotide according to the invention of claim 1 can be used for a forensic analysis.
It is a well-known problem in this technical field to detect further polymorphic sites in human genome DNA.
Moreover, it is a well-known technique in the field to detect polymorphic sites by determining and aligning multiple genomic DNA sequences derived from several persons.
Therefore, it would be easily conceivable for a person skilled in the art to determine multiple human genomic DNA sequences and detect partial DNA sequences which include polymorphic sites which differ by each person.
Furthermore, no unexpected advantageous effect can be acknowledged in relation to the polynucleotides according to the invention of claim 1.
Article 36(4)(i) (Enablement requirement)
In regard to an invention of a product, the description must be stated in such a manner so as to enable a person skilled in the art to produce and use the product.
It is stated in the description that the polynucleotide according to the invention of claim 1 can be used for a forensic analysis. However, a single SNP is usually not sufficient for a forensic analysis. Therefore, the mere statement that it can be used for a forensic analysis is not considered to prove "enabling a person skilled in the art to use" a polynucleotide having the SNP according to the claimed invention.
[Case 15] Case relating to Inventive Step and Enablement Requirements
Title of the invention
SNP
Claims
[Claim 1] A polynucleotide having a consecutive 20-100 bases including the 50 th base in the DNA sequence set forth in SEQ ID NO: 19 (position 50 is g).
Overview of the description
A polynucleotide having the DNA sequence (500 bases in length) set forth in SEQ ID NO: 19 with a substitution of g with c at position 50 has been known.
The 50 th position in the DNA having the sequence set forth in SEQ ID NO: 19 has been shown to be a polymorphic site. In addition, it has been experimentally shown that a polynucleotide having consecutive 20-100 bases including the 50 th base (g) in the DNA sequence set forth in SEQ ID NO: 19 can be used for a diagnosis of disease Z and that such diagnosis of disease Z can be significantly more accurate than conventional methods which use polynucleotides, etc.
No other tools for the diagnosis of disease Z were found to have accuracy as high as that of the diagnosis with the polynucleotides according to the invention of claim 1. [Overview of reason for refusal] None.
(Supplemental explanation)
The polynucleotides according to the invention of claim 1 have a remarkable effect of being available as a tool of highly accurate diagnosis of disease Z.
[Case 16] Case relating to Novelty, Inventive Step and Enablement Requirement
Title of the invention
Partial polypeptide of protein having significantly higher activity
Claims
[Claim 1] An isolated and purified polypeptide consisting of a part of protein P having the amino acid sequence set forth in SEQ ID NO: 1, wherein the part starts with an amino acid selected from a.a. 214 to 218 of the protein P and ends with an amino acid selected from a.a. 394 to 401 of the protein P.
Overview of the description
The protein P has been conventionally known and the amino acid sequence thereof was also conventionally known. It has been also conventionally known that administration of the protein P lowers blood pressure. The inventors found for the first time that the active residues in the binding pocket in the protein P are a.a. 223, 224, 227, 295, 343, 366, 370, 378, and 384. All peptides starting with an amino acid selected from a.a. 214 to 218 and ending with an amino acid selected from a.a. 394 to 401 in SEQ ID NO: 1 were confirmed by X-ray crystallographic analysis to be a protein domain that can be folded into an active binding pocket of the protein P. The peptides consisting of the aforementioned domain alone were demonstrated to exhibit significantly stronger signal activities than the full length protein when activated by a natural ligand of the protein P.
(Supplemental explanation)
The polypeptide has novelty because it can be distinguished from the full length protein (as a substance per se). The prior art teaches neither a polypeptide consisting of a specific part of the protein P nor a method for identifying such a part of the polypeptide. The polypeptide satisfies the requirement for inventive step because it has significantly stronger activity than the full length protein.
[Case 17] Case relating to Inventive Step
Title of the invention
Mutants
Claims
[Claim 1] A polynucleotide encoding a protein selected from the following (i) or (ii):
(i) a protein having an amino acid sequence derived from the amino acid sequence set forth in SEQ ID NO: 1 by substitution of glycine with aspartic acid, alanine, or serine at the 136 th position and having the enzyme A activity; and
(ii) a protein having an amino acid sequence derived from the amino acid sequence set forth in SEQ ID NO: 1 by substitution of valine with leucine, serine, or tyrosine at the 148 th position and having the enzyme A activity.
Overview of the description
A variety of mutants maintaining the enzyme A activity were generated from the protein having the amino acid sequence set forth in SEQ ID NO: 1 and having the enzyme A activity.
The enzyme A activity was improved by substituting glycine with aspartic acid at the 136 th position in the amino acid sequence set forth in SEQ ID NO: 1.
Moreover, glycine at the 136 th position in the amino acid sequence set forth in SEQ ID NO: 1 was substituted with alanine or serine, as well as valine at the 148 th position in the amino acid sequence was substituted with leucine, serine, or tyrosine.
The polynucleotides according to claim 1 provide a variety of mutants having a superior function or an equivalent function of the protein having enzyme A activity and are useful.
(Comparison data has shown that substitution of glycine with aspartic acid at the 136 th position in the amino acid sequence set forth in SEQ ID NO: 1 improves the enzyme A activity of the protein.
It is stated in the description that substitution of glycine with alanine or serine at the 136 th position in the amino acid sequence set forth in SEQ ID NO: 1 improve the enzyme A activity of the protein, but the degree of the improvement was not shown by comparison data or the like.
It can be seen from the comparison data that substitution of valine with leucine, serine, or tyrosine at the 148 th position in the amino acid sequence set forth in SEQ ID NO: 1 did not change the properties such as activity and heat stability of the protein.)
In a case where a protein having a useful function or property is obtained, it was an obvious problem prior to the filing of the application to obtain a protein having a function equivalent or superior to the said protein. As for the techniques to achieve such an object, the techniques of generating mutants by introducing a variety of mutations into the said protein and screening the mutants on the basis of the function or the like to acquire a mutant having a desired function were well-known prior to the filing of the application.
Hence, a person skilled in the art would easily conceive the idea of obtaining mutants having a function equivalent or superior to the protein described in the document 1 based on the aforementioned well-known techniques.
According to the comparison data stated in the description, while substitution of amino acid with aspartic acid at the 136 th position in the amino acid sequence set forth in SEQ ID NO: 1 is found to markedly improve the enzyme A activity of the protein, it is not inferred that substitution of amino acid with alanine or serine other than aspartic acid at the 136 th position in the amino acid sequence set forth in SEQ ID NO: 1 would markedly improve the enzyme A activity, in consideration of the common general knowledge at the time of filing that when amino acid substitutions are introduced into a protein, the influence of the substitutions on a function or the like of the protein varies depending on the type of the amino acid to be introduced even if the amino acid substitutions are introduced at the same position. Furthermore, as of the substitution at the 148 th position in the amino acid sequence set forth in SEQ ID NO: 1, there is no disclosure about an advantageous effect in comparison with the protein described in the document 1.
Therefore, no advantageous effect can be acknowledged on the entire scope of the invention according to claim 1 in view of the description in the document 1. [Measures of the applicant] As for claim 1, once it is asserted based on a certificate of experimental results or the like in a written opinion that both substitution with alanine at the 136 th position and substitution with serine at the 136 th position markedly improve the enzyme A activity of the protein, then deleting (ii) will be sufficient to overcome the reason for refusal. Alternatively, limiting the substitution of amino acid at the 136 th position in the amino acid sequence set forth in SEQ ID NO: 1 to substitution of glycine with aspartic acid and deleting (ii) by amendment will overcome the reason for refusal.
As for amino acid substitution at the 148 th position in SEQ ID NO: 1, even when it is asserted or demonstrated in a written opinion that the substitution has an advantageous effect in comparison with the protein described in document 1, if the effect is not clearly stated in the description and it cannot be inferred from the statement in the description and drawings, then the effect shall not be considered and the reason for refusal shall not be overcome. [Example of a claim that overcome the reason for refusal stated above] [Claim 1] A polynucleotide encoding a protein having the amino acid sequence derived from the amino acid sequence set forth in SEQ ID NO: 1 by substitution of glycine with aspartic acid at the 136 th position.
[Case 18] Case relating to Inventive Step
Title of the invention
Method for assessing genetic risk of non-small-cell lung cancer
Claims
[Claim 1] A method for assessing genetic risk of non-small-cell lung cancer, comprising determining a base of the SNP at the position -50 of gene A (SEQ ID NO: 1), wherein the risk of developing non-small-cell lung cancer is assessed to be high if the nucleotide is G.
Overview of the description
[Background art]
It is known that expression of gene A is increased in lung cancer cell lines and patients. An SNP associated with lung cancer has been identified at the position -50 in the gene A. Lung cancer can be generally classified into non-small-cell lung cancer and small-cell lung cancer depending on the tissue type. The association of the SNP with non-small-cell lung cancer or small-cell lung cancer has not been analyzed. [Problem to be solved by the invention]
An object of the invention is to provide a method for precisely assessing genetic risk of non-small-cell lung cancer, which accounts for about 80% of lung cancer. [Examples]
To evaluate the association of the SNP at the position -50 in the gene A with non-small-cell lung cancer, for the SNP a comparison analysis of a non-small-cell lung cancer patient group and a healthy group was conducted with a comparison analysis of a lung cancer patient group and a healthy group conducted as a Comparative Example.
The odds ratio in the analysis of the non-small-cell lung cancer group was unexpectedly high in comparison with that in the analysis of the lung cancer patient group. In addition, the same analysis of another patient group was conducted and a similar result was obtained. In comparison with known SNPs examined on the association with non-small-cell lung cancer, the SNP of the invention was shown to have a very high correlation with non-small-cell lung cancer.
[Case 19] Case relating to Inventive Step
Title of the invention
Method for assessing genetic risk of non-small-cell lung cancer
Claims
[Claim 1] A method for assessing genetic risk of non-small-cell lung cancer, comprising determining a base of a SNP at the position -50 of gene A (SEQ ID NO: 1), wherein the risk of developing non-small-cell lung cancer is assessed to be high if the nucleotide is G.
Overview of the description
[Background art]
It is known that expression of gene A is increased in lung cancer cell lines and patients. However, no SNP on the gene A was known in association with non-small-cell lung cancer.
[Case 20] Case relating to Inventive Step
Title of the invention
Marker of cancer metastasis
Claims
[Claim 1] A method for identifying a metastatic cancer tissue, comprising the steps of:
(1) detecting presence or absence of mRNA expression transcribed from gene A having the nucleic acid sequence set forth in SEQ ID NO: 1 in a cancer tissue sample from a cancer patient; and
(2) if the cancer tissue sample expresses the mRNA, identifying the cancer tissue sample as metastatic tissue.
Overview of the description
[Background art]
A plurality of genes has been reported to be associated with cancer metastasis, but none of them are sufficiently reliable.
The document 1 discloses that cancer cell lines having high motility and invasive capacity exhibit the expression of gene A and that the gene A is associated with motility and invasive capacity of cancer cells.
The document 1 does not teach the association of the gene A with cancer metastasis itself.
However, it is common general knowledge prior to the filing of the application that cancer cells having higher motility and invasive capacity are more likely to be metastatic.
Therefore, a person skilled in the art would easily conceive the idea of using the presence or absence of mRNA expression transcribed from the gene A as an indicator of cancer metastasis.
Furthermore, unexpectedly advantageous effects over the document 1 cannot be acknowledged for the invention according to claim 1. [Measures of the applicant] The reason for refusal may be overcome by asserting and demonstrating based on a certificate of experimental results or the like in a written opinion that the effect of the invention according to claim 1 that can be inferred from the description could not be predicted from the description in the document 1. For example, the reason for refusal may be overcome by asserting and demonstrating in a written opinion that many of other genes known to be associated with motility and invasive capacity of cancer cells are not useful as a marker of cancer metastasis or that the gene A is superior as a marker of cancer metastasis to other genes which are known to be associated with motility and invasive capacity of cancer cells.
[Case 21] Case relating to Inventive Step
Title of the invention
Method for assessing genetic risk of disease A
Claims
[Claim 1] A method for assessing genetic risk of disease A, comprising determining a base of the SNP at the position 100 in gene A (SEQ ID NO: 1), wherein the risk of developing disease A is assessed to be high if the nucleotide is T.
Overview of the description
[Examples]
To identify SNPs associated with disease A, a comparison analysis of a disease
A patient group and a healthy group was conducted and the SNP (C/T) at the 100 th position in the gene A (SEQ ID NO: 1) was identified.
The documents 2 and 3 disclose that the disease A develops as disease becomes chronic or progresses and disclose examples in which SNPs associated with disease (SNPs in gene B and C) were used for assessing genetic risk of disease A. [Overview of reason for refusal] Article 29(2) (Inventive Step)
The document 1 discloses the SNP(C/T) at the 100 th position in the gene A (SEQ ID NO: 1) as an SNP associated with disease .
Considering that the documents 2 and 3 disclose that the disease A develops as disease becomes chronic or progresses and disclose that SNPs associated with disease can be applied for assessing genetic risk of disease A, a person skilled in the art would easily conceive the idea of using the SNP (C/T) at the 100 th position in the gene A (SEQ ID NO: 1), which is an SNP associated with disease , for assessing genetic risk of disease A.
Furthermore, unexpectedly advantageous effects over the documents 1-3 cannot be acknowledged for the invention according to claim 1.. [Measures of the applicant] The reason for refusal may be overcome by asserting and demonstrating in a written opinion or the like that the effect of the invention according to claim 1 that can be inferred from the description could not be predicted from the description in the documents 1-3. For example, the reason for refusal may be overcome by asserting and demonstrating in a written opinion or the like that the SNP in the gene A of the patent application is superior as a diagnosis marker of disease A compared to SNPs on gene B and C.
[Case 22] Case relating to Inventive Step
Title of the invention
Promoter
Claims
[Claim 1] A polynucleotide having the DNA sequence set forth in SEQ ID NO: 1.
Overview of the description
The polynucleotide having the DNA sequence set forth in SEQ ID NO: 1 is a polynucleotide consisting of 1,370 bases derived from the actinomyces species A of the genus Streptomyces. The polynucleotide was found to locate upstream of the gene encoding enzyme X having the DNA sequence set forth in SEQ ID NO: 2 and to have a promoter activity.
Using a DNA sequence database disclosed prior to the filing of the application, a search was conducted for sequences having a homology with the DNA sequence set forth in SEQ ID NO: 1 and no DNA sequence was found to have an identity equal to or more than 40%.
Thus, the polynucleotide of the invention according to claim 1 encodes a novel promoter and it can be introduced into a host microorganism to produce a protein such as the enzyme X, and therefore it is useful for the production of proteins.
Using a DNA sequence database disclosed prior to the filing of the application, a search was conducted for sequences having a homology with the DNA sequence set forth in SEQ ID NO: 2 and the document 1 was found, which discloses the enzyme X gene from the species B of the genus Streptomyces having an identity of 97% with the DNA sequence.
The species A of the genus Streptomyces is a known microorganism. [Overview of reason for refusal] Article 29(2) (Inventive Step)
In a case where a gene encoding a protein having a useful function or property is obtained from a certain species of organism, it was an obvious problem prior to the filing of the application to obtain a gene encoding a protein having a function equivalent or superior to the said protein and the promoter region of the gene. As for the techniques to achieve such an object, the techniques of obtaining a structural gene of a protein having a corresponding function from a closely related species of organism and cloning a promoter region from the upstream region of the structural gene were well-known prior to the filing of the application.
Therefore, a person skilled in the art would easily conceive the idea of cloning the gene of enzyme X from the known species A belonging to the genus Streptomyces and a promoter region from the upstream region of the gene based on the invention stated in the document 1., Confirming the promoter activity of the promoter region is one of normal practices for a person skilled in the art.
Furthermore, unexpectedly advantageous effects over the document 1 cannot be acknowledged for the invention according to claim 1. [Measures of the applicant] The aforementioned reason for refusal may be overcome by asserting or demonstrating the difficulty in cloning of the polynucleotide of the invention according to claim 1 or a particularly marked effect of high promoter activity etc. of the polynucleotide based on a certificate of experimental results in a written opinion or the like.
[Case 23] Case relating to Inventive Step
Title of the invention
Modified animal
Claims
[Claim 1] A knockout mouse having a loss of function mutation of gene A.
Overview of the description
The description states that a knockout mouse having a loss of function mutation of the gene A was created and examined on its phenotype, and found to have an elevated allergic reaction when exposed to antigen X in comparison with a wild-type mouse. However, no concrete data of comparison experiments indicating the degree of the difference in allergic reaction to antigen X between the knockout mouse and the wild-type mouse to support the statement is shown.
The document 1 discloses, as knockout mice having elevated allergic reactions to the antigen X, knockout mice having a loss of function mutation of gene B or C, both of them involving the suppression of allergic reactions to the antigen X.
The document 2 discloses that expression of gene A suppresses allergic reactions to the antigen X. [Overview of reason for refusal] Article 29(2) (Inventive Step)
It is a well-known problem to create a knockout mouse having a loss of function mutation of a gene implicated in a disease for the purpose of creating a disease model mouse.
In consideration of the description of the document 2, it would be easy for a person skilled in the art to conceive the idea of creating a knockout mouse having a loss of function mutation of the gene A, which has a common property of suppressing the allergic reaction to the antigen X with the genes B and C, instead of a loss of function mutation of the gene B or C in the knockout mouse of the document 1.
Since the description of the application only states that the degree of the allergic reaction to the antigen X in the knockout mouse having the loss of function mutation of the gene A is elevated in comparison with a wild-type mouse, and does not disclose any comparison experimental data indicating the degree of the elevation it is not acknowledged that the invention according to claim 1 has an advantageous effect that cannot be predicted from the documents 1, 2 and the well-known art. [Measures of the applicant] If there was a technical difficulty to create a knockout mouse having a loss of function mutation of the gene A, the reason for refusal may be overcome by asserting the difficulty in a written opinion or the like.
Alternatively, the reason for refusal may be overcome by asserting, based on a certificate of experimental results, etc. in a written opinion or the like, an effect of the claimed invention that can be inferred from the description, such as that "in comparison with the known gene B or C knockout mice exhibiting the phenotype of having elevated allergic reactions to the antigen X, the gene A knockout mouse exhibits markedly higher allergic reactions to the antigen X, and therefore is highly useful as an allergic disease model animal."
[Case 24] Case relating to Inventive Step
Title of the invention
Method for producing differentiated cell from pluripotent stem cell
Claims
[Claim 1] A method for producing a differentiated cell X, comprising the steps of: preparing an embryoid body from a pluripotent stem cell A; and culturing the embryoid body in the presence of substances a, b and c to produce the differentiated cell X.
Overview of the description
[Background art]
It is known that a cell culture containing the differentiated cell X at a purity of 30% can be prepared by culturing an embryoid body obtained from the pluripotent stem cell A in a synthetic medium containing the substances b and c.
When the embryoid body was cultured under similar conditions without the substance a, the content of the differentiated cell X in the cell culture was 20%.
Therefore, the addition of the substance a improved the differentiation efficiency. [Result of the prior art searches] The document 1 discloses that a cell culture containing the differentiated cell X at a purity of 30% was obtained by culturing an embryoid body prepared from the pluripotent stem cell A in a synthetic medium containing the substances b and c for 4 days.
The document 2 discloses that a cell culture containing the differentiated cell X at a purity of 20% was obtained by culturing an embryoid body prepared from the pluripotent stem cell A in a synthetic medium containing the substance a for 3 days. [Overview of reason for refusal] None
(Supplemental explanation)
The document 1 discloses a method for producing the differentiated cell X by culturing an embryoid body derived from the pluripotent stem cell A in the presence of the substances b and c. The document 2 discloses that differentiation into the differentiated cell X is promoted by culturing an embryoid body derived from the pluripotent stem cell A in the presence of the substance a.
However, based on the description of the application, it is admitted that the differentiated cell X can be prepared from an embryoid body at a markedly higher differentiation efficiency by culturing an embryoid body using the substances b and c disclosed in the document 1 and the substance a disclosed in the document 2 in combination. The effect of the invention according to claim 1 is not an effect that a person skilled in the art could predict in consideration of common general knowledge at the time of filing.
[Case 25] Case relating to Novelty and Inventive Step
Title of the invention
Method for producing differentiation cell from pluripotent stem cell
Claims
[Claim 1] A method for producing a differentiated cell X, comprising the steps of: preparing an embryoid body from a pluripotent stem cell A, and culturing the embryoid body in the presence of substances a, b and c to produce the differentiated cell X.
Overview of the description
According to the method of the claimed invention, the differentiation cell X can be produced efficiently at a high purity by culturing an embryoid body in the presence of the substances a, b and c.
Specifically, a cell culture containing the differentiated cell X at a high purity was obtained by culturing the pluripotent stem cell A in a synthetic medium for 2 days to prepare an embryoid body, and then culturing the embryoid body in a synthetic medium containing the substance a, b, and c for 2 days.
However, any concrete data of comparison experiments indicating the degree of the difference in the efficiency of differentiation and the purity of the differentiated cell X between the method of the claimed invention and conventional methods for producing the differentiated cell X is not shown.
The document 2 discloses that differentiation into the differentiated cell X was promoted by treating an embryoid body prepared from the pluripotent stem cell A with the substance a. [Overview of reason for refusal] Article 29(2) (Inventive Step)
Regarding a method for producing a specific differentiated cell by differentiation of a pluripotent stem cell, it is a well-known problem to improve the method so as to produce the differentiated cell efficiently in a shorter time.
The document 1 discloses a method for producing the differentiated cell X by culturing an embryoid body derived from the pluripotent stem cell A in the presence of the substances b and c. The document 2 discloses that differentiation into the differentiated cell X is promoted by treating an embryoid body prepared from the pluripotent stem cell A with the substance a.
Therefore, a person skilled in the art would easily conceive the idea of using substance a at an appropriate concentration to promote differentiation into the differentiated cell X in the method disclosed in the document 1. Although it is stated in the description of the application that the differentiated cell X can be produced more efficiently at a higher purity by the method according to claim 1 than by conventional methods, any data of comparison experiments indicating the degree of improvement in efficiency and purity is not shown. Then, it is not considered that the invention according to claim 1 has a marked effect that a person skilled in the art could not predict. [Measures of the applicant] The reason for refusal may be overcome by asserting based on a certificate of experimental results, etc. in a written opinion or the like an effect of the claimed invention that can be inferred from the description, such as that "in the method according claim 1, the time required for the differentiation into the differentiated cell X can be shortened and the purity of the differentiated cell X obtained is markedly improved in comparison with those in known methods disclosed in the documents 1 and 2."
[Case 26] Case relating to Novelty and Inventive Step
Title of the invention
Method for producing pluripotent stem cell derived from differentiated cell
Claims
[Claim 1] A method for producing a pluripotent stem cell X, comprising introducing genes a, b and c into a gastric epithelium cell.
[Claim 2] A pluripotent stem cell X produced by the method according to claim 1.
Overview of the description
Modified cells in gene expression were produced by introducing the genes a, b and c into gastric epithelial cells, differentiated cells taken from mature tissue. The produced cells exhibited gene expression characteristic to undifferentiated cells and were those which can differentiate into endodermal, mesodermal and ectodermal cells upon induction of differentiation. The efficiency of dedifferentiation was high.
Similarly, the documents 2 and 3 discloses that cells having characteristics of the pluripotent stem cell were produced by introducing the genes a, b and c into a bone marrow-derived cell and a hepatocyte, both derived from mature tissue. [Overview of reason for refusal] Article 29(2) (Inventive step): claim 1
It is a well-known problem to improve a method for producing a useful pluripotent stem cell. Moreover, a method for producing a pluripotent stem cell by introducing the genes a, b and c into a differentiated cell to dedifferentiate it is a well-known technique as disclosed in the documents 1-3.
Therefore, it is an idea a person skilled in the art would conceive easily to apply the aforementioned method disclosed in the documents 1-3, which has been performed with various cells belonging to different histological lines, such as a dermal fibroblast, a bone marrow-derived cell, and a hepatocyte, to a gastric epithelial cell, a cell derived from an another mature tissue, in order to produce the pluripotent stem cell X. Furthermore, unexpectedly advantageous effects over the documents 1-3 and the well-known technique cannot be acknowledged for the invention according to claim 1.
Article 29(1)(iii) (Novelty): claim 2
Since the pluripotent stem cell X of the present application cannot be distinguished from the pluripotent stem cells produced in the documents 1-3, the invention according to claim 2 is not novel in view of the document 1, 2 or 3.
If there is some modifications necessary to successfully apply the method disclosed in the documents 1-3 to the gastric epithelial cell, the reason for refusal may be overcome by making an amendment to include such modifications in the claim and asserting in a written opinion or the like that a person skilled in the art could not conceive the modifications easily.
Alternatively, the reason for refusal may be overcome by asserting based on a certificate of experimental results, etc. in a written opinion or the like that the dedifferentiation efficiency by the method according to claim 1 is markedly higher than that of the method described in the documents 1-3 and the efficiency is an effect that exceeds the scope that can be predicted from the descriptions of the documents 1-3 and the common general knowledge.
Regarding claim 2
The aforementioned reason for refusal regarding novelty shall not be usually overcome unless it is demonstrated that the pluripotent stem cell X of the application is definitely distinguished by objective indicator(s) from the pluripotent stem cells obtained by the method described in the documents 1-3.
(Supplemental explanation)
As of "objective indicator(s)" in "Regarding claim 2" in "Response by the applicant" described above, indicators that can change depending on preservation or culture conditions of the pluripotent stem cell (for example, expression of a specific gene) are not adequate. Such an objective indicator has to be constantly capable of being detected, measured, or observed as a characteristic of the cell (for example, a cell surface marker, the difference in the recombination of a TCR-related gene between iPS cells derived from the T cell and iPS cells derived from the fibroblast). If such an indicator that can be constantly detected, measured, or observed is present, even when it is not indicated in the description, it might be demonstrated by using the indicator that the pluripotent stem cell X of the patent application is different from the pluripotent stem cell described in the documents 1-3.
[Case 27] Case relating to Enablement Requirement
Title of the invention
Monoclonal antibody having a high binding ability to protein A
Claims
[Claim 1] A monoclonal antibody binding to protein A, having a dissociation constant of 10-13 M or more and 10-12 M or less
Overview of the description
The object of the present application is to provide a monoclonal antibody having a high binding ability of 10-13 M or more and 10-12 M or less in dissociation constant against protein A.
As an example, a monoclonal antibody was manufactured using protein A as an immunogen based on the well-known hybridoma method, and a number of hybridomas producing the monoclonal antibody having the biding ability of 10-11 M to 10-9 M in dissociation constant against protein A was obtained. However, as the hybridoma producing the monoclonal antibody having the binding ability of 10-13 M or more and 10-12 M or less in the dissociation constant against protein A, only one strain, called "hybridoma HK" producing the monoclonal antibody having 5.6 10-13 M in the dissociation constant, was obtained.
(Note) The deposition of "hybridoma HK" and its deposited number is not stated in the originally attached description, and a document certifying such a fact is not attached to the request for the application.
[Result of the prior art searches] While the prior art disclosing the monoclonal antibody binding to protein A was found, no prior art disclosing or suggesting the monoclonal antibody binding to protein A with 10-13 M or more and 10-12 M or less in dissociation constant was found. [Overview of reason for refusal] Article 36(4)(i) (Enablement requirement)The description states that only "hybridoma HK" was obtained as a hybridoma producing a monoclonal antibody binding to protein A with 10-13 M or more and 10-12 M or less in the dissociation constant based on the well-known hybridoma method. The deposition of "hybridoma HK" is not stated in the description and it cannot be said that "hybridoma HK" is easily available for a person skilled in the art since "hybridoma HK" is generally commercially available.
In addition, there is a common general knowledge at the time of filing that it is rare that a hybridoma producing a monoclonal antibody complying with a limited condition can be reproducibly obtained. Accordingly, it cannot be readily acknowledged that said hybridoma can be obtained with reproducibility at the time of performing an additional test by a person skilled in the art, since there is no statement in the description that a plurality of strains of hybridomas producing the monoclonal antibody binding to protein A with 10-13 M or more and 10-12 M or less in the dissociation constant were obtained.
Therefore, the description cannot be regarded as stating the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention claimed in Claim 1. [Measures of the applicant] The reason for refusal may be overcome by proving, based on specific and objective evidence in a written opinion or the like, that it is not necessary to make trials and errors, and/or complicated and sophisticated experimentation beyond the extent to which a person skilled in the art should be reasonably expected in order to obtain the monoclonal antibody according to Claim 1 with reproducibility at the time of performing an additional test by a person skilled in the art.
[Case 28] Case relating to Enablement Requirement
Title of the invention
Bifidobacterium bifidum AA strain
Claims
[Claim 1] A Bifidobacterium bifidum AA strain, deposited with accession number NITE P-XXXX.
Overview of the description
As a result of screening for obtaining a new lactic acid bacterium having a function of lowering cholesterol, the Bifidobacterium bifidum AA strain having such a function even in a killed state was isolated.
The taxonomical property of the Bifidobacterium bifidum AA strain was analyzed in detail and the difference with the publicly known strain among the same species was examined. As a result, it was revealed that the Bifidobacterium bifidum AA strain is a new strain.
Hence, the Bifidobacterium bifidum AA strain was deposited with the accession number NITE P-XXXX to NITE-NPMD (National Institute of Technology and Evaluation, NITE Patent Microorganisms Depositary).
The invention according to the above-mentioned Claim relates to the Bifidobacterium bifidum AA strain deposited with accession number of NITE P-XXXX.
In this regard, since the copy of the Receipt of an Original Deposit is not submitted for the AA strain, the fact of deposition cannot be acknowledged. In addition, any circumstance that said strain is easily available for a person skilled in the art, such as being generally commercially available, is not recognized.
Furthermore, it cannot be objectively acknowledged that a person skilled in the art can produce the AA strain without excessive trials and errors and with a high probability, even when the statement in the description of the present application and the common general knowledge at the time of filing are reviewed.
Therefore, the description cannot be regarded as stating the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention claimed in Claim 1. [Measures of the applicant] The reason for refusal will be overcome by submitting the copy of the Receipt of an Original Deposit for the Bifidobacterium bifidum AA strain, deposited with accession number NITE P-XXXX.
(Supplemental explanation)
It is the common general knowledge that it is difficult to obtain a specific strain from any microorganism groups with reproducibility.
Therefore, it is necessary to ensure that a person skilled in the art can easily obtain the specific strain by submitting the copy of the Receipt of an Original Deposit.
[Case 29] Case relating to Enablement Requirement and Support Requirement
Title of the invention
IgM monoclonal antibody to surface antigen P of virus Y
Claims
[Claim 1] An IgM monoclonal antibody, reacting to a surface antigen P of a virus Y with 10-11 M or more and 10-10 M or less in dissociation constant.
[Claim 2] A hybridoma producing a monoclonal antibody according to Claim 1.
Overview of the description
The surface antigen P of the virus Y has been already isolated and purified and the antibody which can detect it had been publicly known at the time of filing.
However, while it has been considered that an IgM monoclonal antibody is not desirable for detection because IgM monoclonal antibodies have an easily aggregable property or the like, the inventors first obtained the IgM monoclonal antibody which can detect the surface antigen P of the virus Y with high sensitivity.
The inventors selected a specific partial amino acid sequence from the amino acid sequence of the surface antigen P, manufactured the polypeptide consisting of the specific partial amino acid sequence, and confirmed that the polypeptide can function as an immunogen. In addition, the hybridomas producing the monoclonal antibodies were manufactured using the polypeptide based on the well-known hybridoma method and 149 strains of hybridoma which produced antibodies were obtained. From them, 10 strains were selected and the dissociation constants of the antibodies which were produced from the selected hybridomas were measured. As a result, 3 strains of hybridoma producing the antibodies which were IgM type with 10-11 M or more and 10-10 M or less in dissociation constant were confirmed.
(Supplemental explanation)
In this case, the monoclonal antibody according to Claim 1 is an IgM monoclonal antibody complying with a limited condition of "reacting to a surface antigen P of a virus Y with 10-11 M or more and 10-10 M or less in dissociation constant".
It is the common general knowledge that to obtain a hybridoma producing a monoclonal antibody complying with a limited condition is often no-replicable.
However, the description states that several strains of the hybridoma producing IgM monoclonal antibodies complying with the limited condition of "reacting to a surface antigen P of a virus Y with 10-11 M or more and 10-10 M or less in dissociation constant" was obtained by selecting a specific partial amino acid sequence from the amino acid sequence of the surface antigen P of the virus Y.
Hence, at the time of performing an additional test by a person skilled in the art, the monoclonal antibody according to Claim 1 and the hybridoma producing it can be obtained with reproducibility.
Therefore, the inventions according to Claims 1 and 2 comply with the enablement requirement.
[Case 30] Case relating to Enablement Requirement and Support Requirement
Title of the invention
Peptide having an agonistic activity against R receptor
Claims
[Claim 1] A peptide having an agonistic activity against R receptor, selected from the group consisting of the following (a) to (c).
(a) a peptide consisting of an amino acid sequence represented by SEQ ID No. 1
(b) a peptide consisting of an amino acid sequence represented by SEQ ID No. 1 in which the amino acid of C terminal is substituted from phenylalanine into tyrosine
(c) a peptide consisting of an amino acid sequence represented by SEQ ID No. 1 in which the amino acid at the second position from the N terminal is substituted from leucine into phenylalanine, isoleucine or tryptophan.
Overview of the description
While several peptides having agonistic activity against R receptor have been known, binding to R receptor was predicted by software for predicting the binding and 50 peptides showing high score were synthesized to test the agonistic activity against R receptor with the aim to produce the peptide having the higher activity. As a result, a peptide having 9 amino acids consisting of the amino acid sequence represented by SEQ ID No. 1 was identified as the peptide showing high agonistic activity against R receptor in comparison with the existing peptide. The peptide strongly and specifically induces the response of R receptor, and can be used for the treatment of a disease relating to the receptor.
While the description of the present application states that the peptide comprising consisting of the amino acid sequence represented by SEQ ID No. 1 shows high agonistic activity against R receptor, it does not specifically state that tests for the agonistic activity against R receptor were performed with variants of the peptide.
Here, upon considering the common general knowledge at the time of filing that it is extremely often that a function of a peptide consisting of short amino acid sequence such as about 9 amino acids is lost even by substituting one amino acid residue, it cannot be inferred that a person skilled in the art can obtain a peptide which has substituted amino acid described in (b) or (c) of Claim 1 of the present application in the amino acid sequence represented by SEQ ID No. 1 and which maintains the agonistic activity against R receptor.
Therefore, the description cannot be regarded as stating the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention claimed in Claim 1.
In addition, upon considering the above-mentioned statement in the description and the common general knowledge at the time of filing, the details provided in the description cannot be expanded or generalized to the scope of the claimed invention.
Therefore, the invention according to Claim 1 goes beyond the extent of disclosure in the description. [Measures of the applicant] The applicant can assert in a written opinion that the description clearly and sufficiently states the invention so as to enable a person skilled in the art to carry out the invention according to Claim 1 and that the details provided in the description can be expanded or generalized to the scope of the invention according to Claim 1 in view of the common general knowledge at the time of filing, by means of, for example, submitting technical document(s) which indicates that it is the common general knowledge at the time of filing that the peptide consisting of 9 amino acids having the agonistic activity against R receptor can maintain its agonistic activity against R receptor even when the amino acid at C terminal or the amino acid at the second position from N terminal is substituted by the specific amino acid described in Claim 1.
If it was determined that the assertion made by the applicant could be accepted, all reasons for refusal would be overcome.
[Case 31] Case relating to Eligibility for Patent
Title of the invention
DNA amplification method, thermal cycler and program for amplifying DNA
Claims
[Claim 1] A DNA amplification method in which is performed by a thermal cycler, using a DNA to be amplified, a primer, a DNA polymerase, and a deoxynucleotide triphosphate (dNTP) which are arranged, comprising the steps of:
(i) a denaturing step of denaturing the DNA to be amplified with X 1 C as a denaturing temperature and X 2 seconds as a denaturing time;
(ii) an annealing step annealing the primer to the denatured DNA to be amplified with Y 1 C as an annealing temperature and Y 2 seconds as an annealing time; and
(iii) an elongating step of elongating the annealed primer by the dNTP and the DNA polymerase with Z 1 C as an elongating temperature and Z 2 seconds as an elongating time to obtain an amplified DNA; wherein the steps of the (i) to (iii) are performed with n cycles.
[Claim 2] A thermal cycler, programmed to perform the method according to Claim 1.
[Claim 3] A program for amplifying DNA, for performing the method according to Claim 1 by the thermal cycler.
The invention according to Claim 1 is a creation of a technical idea utilizing laws of nature, that a DNA to be amplified is denatured with heat, a primer anneals the DNA to be amplified and a complementally chain of the DNA to be amplified is synthesized by utilizing a DNA polymerase and a dNTP. Accordingly, the invention according to Claim 1 is considered as a statutory "invention" in the main paragraph of Article 29(1) ("creation of a technical idea utilizing a law of nature").
Regarding Claims 2 and 3
The inventions according to Claims 2 and 3 are a thermal cycler (an apparatus comprising a computer) performing the method invention according to Claim 1 which is considered as a statutory "invention" and a program for performing the method with the thermal cycler, and they are creations of technical ideas utilizing the laws of nature as a whole. Therefore, the inventions are considered as statutory "invention".
The inventions according to Claims 2 and 3 are the "creation of a technical idea utilizing a law of nature," regardless of utilizing the computer software. Accordingly, the inventions are considered as statutory "invention", without pausing to examine in the terms of the computer software.
[Case 32] Case relating to Eligibility for Patent
Title of the invention
In silico screening method using three-dimensional structural coordinate data, program for performing the method by a computer, and database in which information containing name and structure of a compound identified by the method is recorded
Claims
[Claim 1] A method for identifying a compound which is capable of bind to a protein P, comprising:
a step of applying a three-dimensional molecular modeling algorithm to an atom coordinate of the protein P represented in FIG. 1, for determining space coordinates of a binding pocket of the protein P, and a step of screening in silico a set of space coordinates of stored candidate compounds using the space coordinates of the binding pocket of the protein P.
[Claim 2] A program for performing the method according to Claim 1 by a computer.
[Claim 3] A database storing information containing the names and structures of compounds identified by the method of Claim 1.
Overview of the description
The protein P is a publicly-known protein, and it has been also publicly known that the activation of the protein P decreases blood pressure as well as the amino acid sequence of the protein. While the atomic coordinates of the protein P (raw data of the protein itself in which the ligand is not bound) is represented in FIG. 1, the position of the binding pocket is unclear. The general information for the program predicting the binding pocket of the protein (which outputs a relatively large number of amino acid residues responsible for the binding) is disclosed. In addition, the general information for a program for screening in silico in common use is also disclosed. Furthermore, a docking method by a peptide modeling and a speculative drug design is the well-known fact in this technical field. A person skilled in the art can identify a compound binding to the protein by using a predicting program for the binding pocket and a program for screening in silico.
[Overview of the reason for refusal] Article 29(1), main paragraph (Eligibility for Patent): Claim 3
Mere presentation of information (the feature resides solely in the content of the information, and the main object is to present information), such as presentation per se, means for presentation or a method of presentation, in which a technical feature does not reside ,is not considered as a statutory "invention" in the main paragraph of Article 29(1) ("creation of a technical idea utilizing a law of nature").
What is described in Claim 3 is a database in which the information containing the names and structures of compounds is recorded and has a feature solely in the content of the presented information.
Therefore, what is described in Claim 3 is not considered as a statutory "invention".
(Supplemental explanation) Regarding Claim 1
The matters specifying the invention of "a step of applying a three-dimensional molecular modeling algorithm to an atom coordinate of the protein P indicated in FIG. 1, for determining space coordinates of a binding pocket of the protein P" and " a step of screening in an electronic manner a set of space coordinates of stored candidate compounds using the space coordinates of the binding pocket of the protein P" stated in Claim 1 specify that the invention according to the Claim is to specifically perform the information processing based on the physical nature of the subject. Accordingly, the invention is considered as a statutory "invention" in the main paragraph of Article 29(1).
Regarding Claim 2
The invention according to Claim 2 is a computer program for performing the invention according to Claim 1 which is considered as a statutory "invention", and is the creation of a technical idea utilizing a law of nature in whole. Therefore, the invention according to Claim 2 is considered as a statutory "invention".
The inventions according to Claims 1 and 2 are the "creation of a technical idea utilizing a law of nature", regardless of utilizing a computer software. Accordingly, these inventions are considered as statutory "invention", without pausing to examine in terms of the computer software.
[Case 33] Case relating to Eligibility for Patent
Title of the invention
Three-dimensional structural coordinate date
Claims
[Claim 1] A computer model of a protein P produced by an atomic coordinate stated in FIG. 1. [Claim 2] A data sequence containing an atomic coordinate of a protein P represented by FIG. 1, producing a three-dimensional structure of the protein P when operating based on an algorithm for protein modeling.
[Claim 3] A computer readable record medium in which an atomic coordinate of the protein P represented by FIG. 1 is recorded.
Overview of the description
X-ray crystal structure was analyzed for a crystalline of the protein P which is newly manufactured to obtain a data sequence containing an atomic coordinate stated in FIG. 1. Experimental data and its explanation that the protein P is activated to decrease the blood pressure are indicated in the Example. The algorithm creating a protein model based on the atomic coordinate data was a well-known art at the time of filing. The atomic coordinate of the protein P represented by FIG. 1 is useful for an in silico screening method.
Mere presentation of information (the feature resides solely in the content of the information, and the main object is to present information), such as presentation per se, means for presentation or a method of presentation, in which a technical feature does not reside, is not considered as a statutory "invention" in the main paragraph of Article 29(1) ("creation of a technical idea utilizing a law of nature").
All of the computer model according to Claim 1, the data sequence according to Claim 2 and the computer readable record medium in which the data sequence is stored according to Claim 3 have the characteristic only for the content of information to be presented, and are to mainly present the information. Furthermore, it cannot be said that the presentation of information (presentation per se, means for presentation, a method of presentation and the like) has a technical feature. Accordingly, all of what are described in Claims 1 to 3 are mere presentation of information, and are not the "creation of a technical idea utilizing a law of nature".
Therefore, all of what are described in Claims 1 to 3 are not considered as statutory "invention".
[Case 34] Case relating to Eligibility for Patent
Title of the invention
Pharmacophore
Claims
[Claim 1] A pharmacophore having an atomic spacial arrangement in a molecule, defined by the following formula:
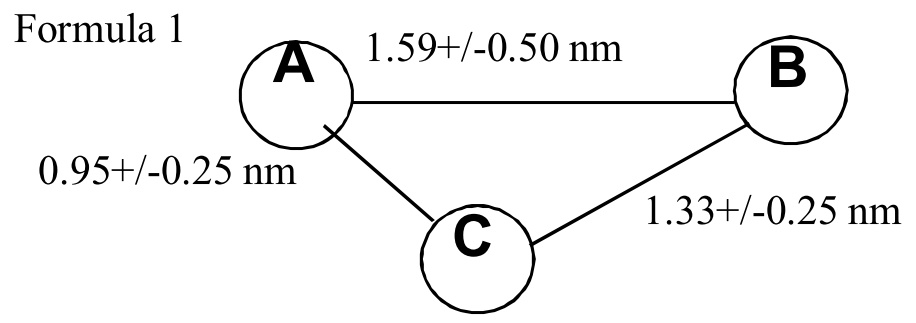
wherein A and B indicate an electron donor group, C indicates a carbon atom constituting a part of a hydrophobic group, and distances indicate a distance between the centers of the atoms.
Overview of the description
In the present application, the pharmacophore is of expressing a comprehensive concept of the molecular characteristic which is expressed by the terms particular to the information for the spacial arrangement of the chemical elements (for example, hydrophobic group, charged / ionic group, hydrogen-bond donor group / accepting group, molecular basic skeleton) which are considered to be responsible for the desired biological activity. The protein P has been a publicly known protein and its amino acid sequence has been also publicly known. It has been publicly known that the activation of the protein P decrease blood pressure. In the pharmacophore represented by [Formula 1], the structure of the ligand binding pocket of the protein P is determined according to the three-dimensional structure of the ligand binding pocket of the protein P which is predicted by a routine method. A novel ligand is designed based on the pharmacophore, and the ligand can bind to the protein with a relatively high affinity.
Mere presentation of information (the feature resides solely in the content of the information, and the main object is to present information), such as presentation per se, means for presentation or a method of presentation, in which a technical feature does not reside ,is not considered as a statutory "invention" in the main paragraph of Article 29(1) ("creation of a technical idea utilizing a law of nature").
The pharmacophore does not mean a substance, its technical feature is only present in the content of information and the pharmacophore itself is nothing more than mere presentation of information.
Therefore, what is described in this Claim is not considered as a statutory "invention".
[Case 35] Case relating to Novelty, Inventive Step and Enablement Requirements
Title of the invention
Crystal of protein
Claims
[Claim 1] Crystal of a protein P, having unit lattice constants of a=4.0 nm, b=7.8 nm and c=11.0 nm.
Overview of the description
The amino acid sequence of the protein P was publicly known. It has been also publicly known that the administration of the protein P decreases the blood pressure. The present inventors succeeded in newly manufacturing a stable crystal of the protein P. The method of manufacturing the crystal is stated in the description and the experimental data. While the protein P in the crystal form is inactive, to regain its activity again by dissolving the crystal into a solution is also proved by the experimental data. It is also proved by the experimental data that the routine prior art used for the crystallization of a protein cannot be applied to the protein P, and it is obvious that there has been technical difficulties in manufacturing the claimed crystal of the protein P.
In addition, there was no prior art relating to a method of crystallizing the protein P. [Overview of the reason for refusal] None.
(Supplemental explanation)
The invention of the crystal of the protein P has novelty since a crystal of a protein can be distinguished and be different from a protein which is not crystallized in terms of its shape and structure. The prior art does not teach the crystal of the protein P or the method of manufacturing the crystal of the protein P stated in the Claim.
Further, the crystallization of the protein P was not successfully achieved by the publicly known method used for crystallizing a protein. Therefore, the invention according to the above-mentioned crystal involves an inventive step.
[Case 36] Case relating to Novelty
Title of the invention
In silico screening method using coordinate data of co-crystal and three-dimensional structure of protein
Claims
[Claim 1] A co-crystal of a protein P and a ligand Q, having unit lattice constants of a=4.0 nm, b=7.8 nm and c=11.0 nm.
[Claim 2] A method for identifying a compound binding to a protein P by comparing a three-dimensional structure of a candidate compound with a three-dimensional molecular model represented by FIG. 5, comprising the steps of:
(1) ...
(2) ...
(...) ...
(n) ...
(the three-dimensional molecular model of FIG. 5 indicates positions of hetero atoms which are contained in amino acids constituting a binding pocket of the protein P (that is, amino acids 223, 224, 227, 295, 343, 366, 370, 378 and 384) and which can form a hydrogen bond with a hydrogen binding functional group of the candidate compound.
The steps (1) to (n) are a data processing method summarized as follows; a) the coordinate data of the three-dimensional molecular model represented by FIG. 5 is input to a data structure such that the distance between atoms of the protein P can be easily retrieved; b) a distance between the hetero atom forming the binding pocket in the three-dimensional molecular model and the hydrogen binding functional group of different candidate compounds is compared, whereby the candidate compound theoretically constituting a most stable complex can be identified by the three-dimensional molecular model of the binding pocket of the protein P based on an optimal hydrogen bond between these two structures.)
Overview of the description
The protein P had been a publicly known protein and its amino acid sequence had been also publicly known. It had been also publicly known that administration of the protein P decreases the blood pressure. The inventors were succeeded in newly manufacturing a stable co-crystal of the protein P and a natural ligand Q. The atomic coordinate in the co-crystalized state of the protein P and the natural ligand Q was defined as a result of X-ray crystal structure analysis. It could be concluded that active amino acid residues of the binding pocket of the protein P are amino acids 223, 224, 227, 295, 343, 366, 370, 378 and 384. It was also disclosed how the three-dimensional molecular model represented by FIG. 5 contains the three-dimensional structure of the binding pocket of the protein P. Several compounds have been identified in the example by the claimed identification method. It was confirmed that the identified compounds are actually bound to the protein P and their binding affinities are indicated as experimental data. It was presumed that several compounds which are strongly bound to the protein P to such a degree that some biological activity can be expected may be actually identified by the method as claimed based on the provided data.
The novelty for the invention according to Claim 1 is acknowledged.
Where the content of the data as a difference between the claimed invention and the prior art is given, the novelty for the claimed invention by this difference shall not be acknowledged (see 2.2.4(1) in "Appendix C Chapter 1 Computer software related invention"). In this case, the difference of the subject to be processed of "three-dimensional molecular model represented by FIG. 5" given as the difference between the method of identifying the compound disclosed in the prior art and the method of identifying the compound stated in Claim 2 does not affect to change the procedure of the information processing of the computer. In addition, this difference is just characterized only by the content of the data. Therefore, the novelty for the invention according to Claim 2 is not acknowledged by this difference. [Measures of the applicant] The above-mentioned reason for refusal relating to novelty for the invention according to Claim 2 may be overcome by an amendment of adding an actual experimental step not disclosed in the prior art as a pre-treatment step or a post-treatment step of the "step of identifying the compound binding to the protein P in silico".
For example, the above-mentioned reason for refusal for novelty may be overcome by amending it into a method comprising the actual experimental step and the step of identifying the compound, wherein it is a method performing a "step of identifying the compound binding to the protein P in silico," after, as the pre-treatment step, the co-crystal according to Claim 1 is manufactured, and the step of identifying the atomic coordinate of the co-crystal is performed by X-ray crystal structure analysis.
Alternatively, the above-mentioned reason for refusal for novelty may be overcome by amending it into a method comprising the step of identifying the compound and the actual experimental step, wherein it is a method performing the "step of identifying the compound binding to the protein P in silico," followed by performing the step of assaying whether or not the compound identified in silico is actually bound to the protein P in vitro, etc. as the post-treatment step.
[Case 37] Case relating to Enablement Requirement, Support Requirement and Clarity Requirement
Title of the invention
Compound identified by in silico screening method
Claims
[Claim 1] A compound identified by a method for identifying a compound binding to a protein P by comparing a three-dimensional structure of a candidate compound with a three-dimensional molecular model represented by FIG. 5, the method comprising the steps of:
(1) ...
(2) ...
(...) ...
(n) ...
(the three-dimensional molecular model of FIG. 5 indicates positions of hetero atoms which are contained in an amino acids constituting a binding pocket of the protein P (that is, amino acids 223, 224, 227, 295, 343, 366, 370, 378 and 384) and which can form a hydrogen bond with a hydrogen binding functional group of the candidate compound.
The steps (1) to (n) are a data processing method summarized as follows; a) the coordinate data of the three-dimensional molecular model represented by FIG. 5 is input to a data structure such that the distance between atoms of the protein P can be easily retrieved; b) a distance between the hetero atom forming the binding pocket in the three-dimensional molecular model and the hydrogen binding functional group of different candidate compounds is compared, whereby the candidate compound theoretically constituting a most stable complex can be identified by the three-dimensional molecular model of the binding pocket of the protein P based on an optimal hydrogen bond between these two structures.)
Overview of the description
The protein P had been a publicly known protein and its amino acid sequence had been also publicly known. It had been also publicly known that the administration of the protein P decreases the blood pressure. The atomic coordinate in the co-crystalized state of the protein P and the natural ligand Q was defined as a result of X-ray crystal structure analysis. It could be concluded that active amino acid residues of the binding pocket of the protein P are amino acids 223, 224, 227, 295, 343, 366, 370, 378 and 384. It was also disclosed how the three-dimensional molecular model represented by FIG. 5 contains the three-dimensional structure of the binding pocket of the protein P. The compound X was identified in the example by the claimed identification method. The actual binding affinity of the identified compound X was showed as the experimental data. It was presumed that several compounds which are strongly bound to the protein P to such a degree that some biological activity can be expected may be actually identified by the method as claimed based on the provided data.
While Claim 1 comprehensively states the compound specified by the identification method using the three-dimensional molecular model represented by FIG. 5, the description only states the compound X as a specific example, and does not specifically state another compound of the compound according to Claim 1. In light of the common general knowledge at the time of filing that it is difficult to understand what the compound defined only by the in silico screening method is specifically, no basis can be found for expanding or generalizing the details provided in the description to the scope of the invention according to Claim 1 defined only by the identification method using the three-dimensional molecular model represented by FIG. 5.
Therefore, the invention according to Claim 1 goes beyond the extent of disclosure in the description.
In addition, since it cannot be understood what the compound according to
Claim 1 except the compound X is specifically, in consideration of the above-mentioned statement in the description and the common general knowledge at the time of filing, it is determined to be necessary to make trials and errors that a myriad of compounds are screened and examined for their binding affinities upon practicing the invention according to Claim 1 beyond the extent to which a person skilled in the art should be reasonably expected.
Therefore, the description does not state the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention according to Claim 1.
Article 36(6)(ii) (Clarity Requirement)
In light of the common general knowledge at the time of filing that it is difficult to understand what the compound defined only by the in silico screening method is specifically, it is obvious that the "compound" in which its chemical structure etc. are not specified and which is defined only by the above-mentioned screening method is not technically and sufficiently specified. In addition, even in consideration of the statement in the description and drawings, the invention cannot be clearly understood based on the statement in the Claim 1.
For example, if the Claim 1 is amended as follows, all of the reasons for refusal will be overcome.
[Claim 1] A compound X binding to a protein P.
[Case 38] Case relating to Enablement Requirement, Support Requirement and Clarity Requirement
Title of the invention
A compound defined by pharmacophore
Claims
[Claim 1] An isolated compound or a salt thereof, defined by a pharmacophore having a spacial arrangement of atoms in a molecule, defined by the following formula.
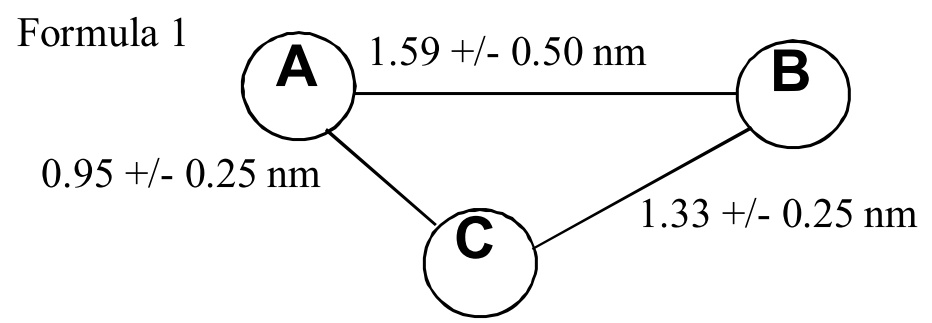
wherein A and B indicate electron donor groups, C indicates a carbon atom constituting a part of a hydrophobic group, and distances indicate a distance between the centers of atoms.
Overview of the description
In the present application, the pharmacophore is of expressing a comprehensive concept of the molecular characteristic which is defined by the expression particular to the information for the spacial arrangement of chemical elements (for example, hydrophobic group, charged / ionic group, hydrogen-bond donor group / accepting group, molecular basic skeleton) which are considered to be responsible for the desired biological activity. The protein P has been a publicly known protein and its amino acid sequence has been also publicly known. It has been publicly known that the activation of the protein P decreases blood pressure. The pharmacophore represented by [Formula 1] was determined based on the three-dimensional structure of the ligand binding pocket of the protein P predicted by a routine method. A novel ligand Q which was designed based on the pharmacophore was synthesized, and it was proved that the ligand binds to the protein P with a relatively high affinity.
While Claim 1 comprehensively states the compounds which are defined by the properties and positions of three atoms, the description only states the novel ligand Q as a specific example, and does not specifically state another compound according to Claim 1. In light of the common general knowledge at the time of filing that it is difficult to understand the ligand structure only by defining the properties and the positions of only three atoms, no basis can be found for expanding or generalizing the details provided in the description to the scope of the invention according to Claim 1 defined only by the pharmacophore represented by Formula 1.
Therefore, the invention according to Claim 1 goes beyond the extent of disclosure in the description.
In addition, in consideration of the above-mentioned statement in the description and the common general knowledge at the time of filing, the ligand structure represented by Formula 1 cannot be understood except the ligand Q indicated in the example. Accordingly, it is determined to be necessary to make trials and errors that a myriad of compounds are screened and examined for their binding affinities upon practicing the invention according to Claim 1 beyond the extent to which a person skilled in the art should be reasonably expected.
Therefore, the description does not state the invention clearly and sufficiently so as to enable a person skilled in the art to carry out the invention according to Claim 1.
Article 36(6)(ii) (Clarity Requirement)
In light of the common general knowledge at the time of filing that it is difficult to understand the ligand structure complying with such a definition only by defining the properties and the positions of only three atoms, it is obvious that the "compound" in which no chemical structure etc. are defined and which is defined only by the properties and the positions of only three atoms is not technically and sufficiently specified. Even when taking into account the statement in the description and drawings, the invention cannot be clearly understood based on the statement in the Claim 1.
For example, if the Claim 1 is amended as follows, all of the reasons for refusal will be overcome.
[Claim 1] A ligand Q or a salt thereof binding to a protein P.
6.2 Cases relating to Determination of Necessity for Deposit of Microorganisms, etc.
[Case 39] Case in which the microorganism is easily available for a person skilled in the art (no necessity for deposit)
Title of the invention
-galactosidase
Claims
[Claim 1] A -galactosidase which is derived from Streptomyces lividans xyz-1 strain
(ATCC ******), having the following physicochemical properties:
(a) action and substrate specificity: hydrolyzing a substrate having -D-galactoside bond to release a D-galactose group.
(b) optimal pH: 4.5
(c) stable pH: 3.0 to 5.5
(d) optimal temperature: 55 ℃
(e) stable temperature: 50 ℃
(f) molecular weight: 200 kD as measured by gel permeation method.
Overview of the description
While it is desired to obtain -galactosidase having a sufficient enzymatic activity at the acidic region since raw materials of neutral to acidic pH such as milk, cheese whey, lactose solution are assumed as subjects to be processed by -galactosidase, microorganisms producing -galactosidase having the sufficient enzymatic activity in the acidic region were not known at the time of filing the present application.
The present inventors isolated the -galactosidase according to Claim 1 from Streptomyces lividans xyz-1 strain by a specific approach. In addition, the Streptomyces lividans xyz-1 strain was listed as storage number ATCC ****** in the catalog published from ATCC, and could be freely furnished prior to filing the present application.
Accordingly, the Streptomyces lividans xyz-1 strain is a microorganism which is easily available for a person skilled in the art and thus a person skilled in the art can isolate the -galactosidase according to Claim 1 by using the specific approach stated in the description.
Therefore, it is not necessary to deposit the Streptomyces lividans xyz-1 strain.
[Case 40] Case in which bacteria are not easily available for a person skilled in the art (necessary to deposit)
Title of the invention
Dioxin degrading bacterium
Claims
[Claim 1] A Bacillus subtilis T-169 strain, having dioxin decomposing ability.
Overview of the description
Saline mud at Toyama Bay was collected as a sample, and the Bacillus subtilis T-169 strain was isolated from the sample by a well-known method for a person skilled in the art. The taxonomical property of the Bacillus subtilis T-169 strain was analyzed in detail and the difference with the publicly known bacteria strain among the same species was examined. As a result, it was found that the Bacillus subtilis T-169 strain is a new bacteria strain. In addition, it was revealed upon performing experiments that the Bacillus subtilis T-169 strain can decompose dioxin with high efficacy.
Accordingly, even where a new microorganism is isolated using a sample collected from the soil, sea water, and the like in the specific region, it is difficult to obtain the new microorganism with reproducibility, as long as there is no reasonable basis that the new microorganism is present in the sample which is re-collected from the soil, sea water and the like.
In this case, the description does not provide a reasonable basis that the Bacillus subtilis T-169 strain is present in the sample which is re-collected from the saline mud at Toyama Bay.
Hence, since the Bacillus subtilis T-169 strain cannot be obtained with reproducibility when a person skilled in the art performs an additional test, the Bacillus subtilis T-169 strain is not a microorganism which can be manufactured by a person skilled in the art based on the statement in the description.
Therefore, it is necessary to deposit the Bacillus subtilis T-169 strain, since the Bacillus subtilis T-169 strain is not a microorganism which is easily available for a person skilled in the art.
[Case 41] Case of the invention according to DNA derived from bacteria (no necessity for deposit)
Title of the invention
DNA derived from bacteria
Claims
[Claim 1] DNA encoding argininosuccinic acid synthase derived from coryneform bacterium strain K-336 and containing a nucleotide sequence represented by SEQ ID No. 1.
[Claim 2] An expression vector comprising the DNA according to Claim 1.
[Claim 3] A transformant bearing the expression vector according to Claim 2.
Overview of the description
The taxonomical property of the coryneform bacterium strain K-336 producing L-arginine which was isolated from the soil based on chemical tolerance was analyzed in detail to examine any variation with native similar species. As the result, it was revealed that the coryneform bacterium strain K-336 is a new species.
It was publicly known prior to the filing the present application that the group of genes including ArgA gene and ArgH gene is responsible for L-arginine biosynthesis pathway in the coryneform bacterium. The inventors first isolated and purified ArgG gene containing the nucleotide sequence represented by SEQ ID No. 1 from the coryneform bacterium strain K-336, and expressed ArgG gene by well-known gene engineering approaches and they have confirmed that a protein encoded by ArgG gene is argininosuccinic acid synthase.
Therefore, it is not necessary to deposit the coryneform bacterium strain K-336.
[Case 42] Case in which a person skilled in the art can manufacture a hybridoma based on the statement in the description (no necessity for deposit)
Title of the invention
Antigen protein A
Claims
[Claim 1] An antigen protein A consisting of an amino acid sequence represented by SEQ ID No. 1.
[Claim 2] A monoclonal antibody to the antigen protein A according to Claim 1.
[Claim 3] A hybridoma producing the monoclonal antibody according to Claim 2.
Overview of the description
A new antigen protein A was isolated and purified from an outer membrane of a virus X. Since the antigen protein A reacts only with serum derived from a person infected with the virus X, the antigen protein A is useful for identifying people infected with the virus X.
In addition, a partial amino acid sequence of the antigen protein A was determined and a gene encoding the antigen protein A consisting of an amino acid sequence represented by SEQ ID No. 1 was cloned by well-known gene engineering approaches based on the partial amino acid sequence.
(Note) There is no example of producing a monoclonal antibody specifically reacting to the antigen protein A.
[Explanation relating to determination of necessity for deposit of microorganisms, etc.] In this case, the monoclonal antibody according to Claim 2 is a monoclonal antibody specified only by an antigen.Generally, when a protein having immunogenicity is obtained, it is the common general knowledge that a monoclonal antibody against the protein can be obtained by the well-known hybridoma method using the protein as an immunogen.
In addition, based on the statement in the description, a person skilled in the art can obtain a gene encoding the antigen protein A, express the gene using a well-known gene engineering approach and prepare the antigen protein A consisting of the amino acid sequence represented by SEQ ID No. 1. Furthermore, it is obvious that the antigen protein A has immunogenicity.
Hence, based on the statement in the description, a person skilled in the art can prepare the antigen protein A, and can obtain a monoclonal antibody according to Claim 2 and a hybridoma producing the same by the well-known hybridoma method using the antigen protein A as an immunogen.
Accordingly, the hybridoma according to Claim 3 is a microorganism which can be manufactured by a person skilled in the art based on the statement in the description.
Therefore, it is not necessary to deposit the hybridoma according to Claim 3 since the hybridoma according to Claim 3 is a microorganism which is easily available for a person skilled in the art.
[Case 43] Case in which a person skilled in the art can manufacture a hybridoma based on the statement in the description (no necessity for deposit)
Title of the invention
A monoclonal antibody
Claims
[Claim 1] An IgM monoclonal antibody, reacting to a surface antigen P of a virus Y with 10-10 M or less in dissociation constant.
[Claim 2] A hybridoma producing the monoclonal antibody according to Claim 1.
Overview of the description
The surface antigen P of the virus Y has already been isolated and purified, and the antibody which can detect the same was publicly known prior to the filing the present application. However, while it has been considered that an IgM monoclonal antibody is not desirable for detection because the IgM monoclonal antibodies have an easily aggregable property or the like, the inventors have first obtained the IgM monoclonal antibody which can detect the surface antigen P of the virus Y with high sensitivity.
The inventors selected a specific partial amino acid sequence from an amino acid sequence of the surface antigen P, manufactured a polypeptide consisting of the specific partial amino acid sequence and confirmed that the polypeptide can function as an immunogen. Further, they manufactured hybridomas producing monoclonal antibodies by the well-known hybridoma method using the polypeptide. As a result, 149 hybridoma strains which are confirmed to produce antibodies were obtained.
When 10 strains were selected from thereamong and binding constants of the antibodies produced from the selected hybridomas were measured, 3 hybridoma strains producing the IgM type antibodies with 10-10 M or less in the dissociation constant were confirmed.
Generally, it is the common general knowledge that a hybridoma producing a monoclonal antibody complying with a limited condition is obtained with almost no reproducibility.
However, the description states that multiple hybridoma strains producing the IgM monoclonal antibodies complying with the limited condition of "reacting to a surface antigen P of a virus Y with 10-10 M or less in dissociation constant" were obtained by selecting the specific partial amino acid sequence from the amino acid sequence of the surface antigen P of the virus Y.
Hence, when a person skilled in the art performs an additional test, the monoclonal antibody according to Claim 1 and the hybridoma producing the same can be obtained with reproducibility.
Accordingly, the hybridoma according to Claim 2 is a microorganism which can be manufactured by a person skilled in the art based on the statement in the description.
Therefore, it is not necessary to deposit the obtained hybridoma since the hybridoma according to Claim 2 is a microorganism which is easily available for a person skilled in the art.
[Case 44] Case in which a hybridoma is not easily available for a person skilled in the art (necessary to deposit)
Title of the invention
A monoclonal antibody
Claims
[Claim 1] A monoclonal antibody ABC-1, binding to a receptor Z to suppress cell growth.
[Claim 2] A hybridoma H-ABC-1 producing the antibody according to Claim 1.
Overview of the description
The receptor Z has already been isolated and purified, and it was publicly known prior to the filing the present application that an agonist binds to the receptor Z to suppress cell growth. In addition, several trials to manufacture a monoclonal antibody binding to the receptor Z to suppress cell growth had already been made as of filing. However, an antibody binding to the receptor Z to suppress cell growth has not been obtained as of filing.
The inventors manufactured monoclonal antibodies by the well-known hybridoma method using the receptor Z as an immunogen. A number of hybridomas producing monoclonal antibodies binding to the receptor Z were obtained, but the hybridoma producing the monoclonal antibody which suppresses cell growth was only one strain thereamong. The monoclonal antibody suppressing cell growth was named as a "monoclonal antibody ABC-1" and the hybridoma producing the "monoclonal antibody ABC-1" was named as a "hybridoma H-ABC-1".
Generally, it is the common general knowledge that it is difficult to intentionally obtain a specific hybridoma strain by the well-known hybridoma method.
Besides, the description states that only one strain of the hybridoma H-ABC-1 producing the monoclonal antibody ABC-1 was obtained based on the well-known hybridoma method, and does not state a method for obtaining the hybridoma H-ABC-1 with reproducibility.
Hence, the hybridoma H-ABC-1 is not a microorganism which can be manufactured by a person skilled in the art based on the statement in the description since the monoclonal antibody ABC-1 or the hybridoma H-ABC-1 cannot be obtained with reproducibility when a person skilled in the art performs an additional test.
Therefore, it is necessary to deposit the hybridoma H-ABC-1 since the hybridoma H-ABC-1 is not a microorganism which is easily available for a person skilled in the art,
[Case 45] Case in which a person skilled in the art can manufacture cells based on the statement in the description (no necessity for deposit)
Title of the invention
Lung cancer cells
Claims
[Claim 1] A method for separating lung cancer cells derived from mouse from an inhomogeneous cell population containing the lung cancer cells derived from mouse, comprising the steps of:
(1) preparing a vector bearing a nucleic acid molecule encoding a fluorescence protein, linked under the control of a lung cancer cell-specific promoter consisting of a nucleotide sequence represented by SEQ ID No. 1;
(2) introducing the vector into the cell population; and
(3) identifying and separating the lung cancer cells derived from mouse among the cell population as cells emitting fluorescence.
[Claim 2] Lung cancer cells derived from mouse, separated by the method according to Claim 1.
Overview of the description
A new promoter specifically functioning in lung cancer cells was cloned from mouse. The nucleotide sequence of the promoter is represented by SEQ ID No. 1. In addition, the inhomogeneous cell population containing the lung cancer cells was prepared from mouse based on the well-known art. Next, a vector bearing a nucleic acid molecule encoding GFP which is well-known as one of fluorescence proteins was introduced under the control of the promoter such that GFP was expressed only in the lung cancer cells among the cell population, and the lung cancer cells derived from mouse were identified and separated as cells emitting fluorescence among the cell population.
Hence, when a person skilled in the art performs an additional test, the lung cancer cells derived from mouse can be identified and separated with reproducibility.
Accordingly, the lung cancer cells derived from mouse according to Claim 2 are microorganisms which can be manufactured by a person skilled in the art based on the statement in the description.
Therefore, it is not necessary to deposit the identified and separated lung cancer cells derived from mouse since the lung cancer cells derived from mouse according to
Claim 2 are microorganisms which are easily available for a person skilled in the art,.
[Case 46] Case in which cells are not easily available for a person skilled in the art (necessary to deposit)
Title of the invention
Mesenchymal stem cells
Claims
[Claim 1] A mesenchymal stem cell strain H01, which is derived from mouse mesenchymal stem cells, can be subcultured in a serum-free medium, shows a fibrous form upon culturing the serum-free medium, and is induced differentiation into cells of interest at a rate of 80% or more by culturing it in a medium containing a conditioned medium for the cells of interest.
Overview of the description
The mesenchymal stem cells obtained from mouse bone marrow were cultured in serum-free culture medium for 3 weeks to remove the killed cells. Thereafter, while the passage for the remained cells was repeated to examine the differentiating ability, a mutant cell strain differentiating into astrocyte like cells by culturing it in the medium containing a conditioned medium for astrocyte was incidentally obtained. The mutant cell strain was named as a mesenchymal stem cell strain H01. When the mesenchymal stem cell strain H01 was further analyzed for the differentiating ability, the strain H01 was induced differentiation into each cell at a rate of almost 100% by culturing it in a medium containing a conditioned medium for adipose cells, smooth muscle cells, fibroblasts and the like.
In this case, the description merely states that the mesenchymal stem cell strain
H01 was established from the mutant cell strain incidentally obtained during the process of subculturing the mesenchymal stem cells derived from mouse bone marrow, and does not state a method for obtaining the mesenchymal stem cell strain H01 with reproducibility.
Accordingly, since the mesenchymal stem cell strain H01 cannot be obtained with reproducibility when a person skilled in the art performs an additional test, the mesenchymal stem cell strain H01 is not a microorganism which can be manufactured by a person skilled in the art based on the statement in the description.
Therefore, it is necessary to deposit the mesenchymal stem cell strain H01 since the mesenchymal stem cell strain H01 is not a microorganism which is easily available for a person skilled in the art.
[Case 47] Case in which a person skilled in the art can manufacture an animal based on the statement in the description (no necessity for deposit)
Title of the invention
Transgenic mouse
Claims
[Claim 1] A transgenic mouse, in which a proto-oncogene consisting of a nucleotide sequence represented by SEQ ID No. 1 is introduced.
Overview of the description
A new proto-oncogene consisting of the nucleotide sequence represented by
SEQ ID No. 1 was cloned from human. Further, the gene was introduced into a commercially available fertilized ovum of BALB/C mouse based on the well-known gene introduction method to prepare a plurality of transgenic mice. As a result, tumors occurred at 5 months age in these mice after birth on average.
Hence, when a person skilled in the art performs an additional test, the transgenic mouse in which the proto-oncogene consisting of the nucleotide sequence represented by SEQ ID No. 1 is introduced can be prepared with reproducibility.
Accordingly, the transgenic mouse according to Claim 1 is an animal which can be manufactured by a person skilled in the art base on the statement in the description.
Therefore, it is not necessary to deposit the prepared transgenic mouse (its fertilized ovum and the like) since the transgenic mouse according to Claim 1 is an animal which is easily available for a person skilled in the art.
[Case 48] Case in which an animal is not easily available for a person skilled in the art (necessary to deposit)
Title of the invention
A mutant mouse
Claims
[Claim 1] An RFG mouse, in which dermatitis spontaneously occurs, having a property that edema is found around the eye at 3 weeks age after birth as a primary lesion.
Overview of the description
During the process of maintaining BALB/c mouse line, a mutant mouse in which edema is found around the eye at 3 weeks age after birth as a primary lesion and in which dermatitis spontaneously occurs under a clean environment was incidentally obtained. Thereafter, an inbred line was established from the mutant mouse and named as an RFG mouse. After establishing the inbred line, during the process of passing 25 generations, the RFG mouse maintained a property that edema is found around eye at 3 weeks of age after birth as the primary lesion and dermatitis spontaneously occurs.
In this case, the description merely states that the RFG mouse was an inbred line which was established from the mutant mouse incidentally obtained during the process of maintaining BALB/c mouse line, and does not state a method for obtaining the RFG mouse with reproducibility.
Accordingly, since the RFG mouse cannot be obtained with reproducibility when a person skilled in the art performs an additional test, the RFG mouse is not an animal which can be manufactured by a person skilled in the art based on the statement in the description.
Therefore, it is necessary to deposit the RFG mouse (its fertilized ovum and the like) since the RFG mouse is not an animal which is easily available for a person skilled in the art.